The National Institute for Health and Care Excellence (NICE) issued medical technology guidance recommending the use of Boston Scientific Corporation cardiac resynchronization therapy defibrillators (CRT-D) powered by EnduraLife™ Battery Technology for treating patients with heart failure.
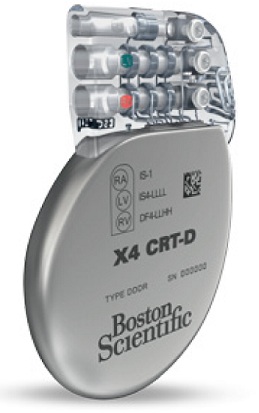
In their evaluation, NICE which provides evidenced-based guidance to improve health and social care in the National Health Service (NHS) in the United Kingdom concluded that the extended battery life observed with Boston Scientific EnduraLife-powered CRT-Ds is likely to reduce the number of avoidable replacement procedures a patient may have to undergo, thereby offering improved outcomes for patients and potential savings to the NHS in England of approximately £6 million in the first five years.
The savings were calculated by modeling the costs associated with early replacement procedures, such as a reduction in hospital admissions, bed days and procurement costs. Fewer replacement procedures also equate to a reduction in associated costs accrued from post-operative complications and infections both of which also have a measurable impact on morbidity and mortality.
“The NICE guidance reinforces the multi-faceted importance of battery longevity in devices treating patients with heart failure and life-threatening ventricular arrhythmias,” said Dr. Jay Wright, consultant cardiologist, Liverpool Heart and Chest Hospital, Liverpool, United Kingdom. “Additionally, the reduction in replacement procedures and potential savings identified within the guidance could offer longer-term relief to NHS providers who have recently seen an increase in the number of patients requiring further in-hospital treatment.”
The NICE medical technologies advisory committee found that the evidence from 16 independent clinical and economic studies supported a case for adoption and concluded that EnduraLife-powered CRT-Ds have a greater battery capacity and longer battery life compared with other CRT-Ds reviewed.
“Battery life should be a key consideration in cardiac device selection for the millions of patients who rely on them daily,” said Kenneth Stein, M.D., senior vice president and chief medical officer, Global Health Policy and Rhythm Management, Boston Scientific. “We are proud to have our EnduraLife Battery Technology recognized by NICE’s evidence-based review process and available for the benefit of patients and the healthcare system at large.”
EnduraLife Battery Technology, which has nearly two times the usable battery capacity as certain competitive devices[i],[ii] and is packaged in a device up to 18 percent smaller than other CRT-Ds[iii],[iv],[v], was introduced in the company’s line of CRT-Ds in 2008.
For more information on the NICE guidance, visit https://www.nice.org.uk/guidance/mtg33.
About Boston Scientific
Boston Scientific transforms lives through innovative medical solutions that improve the health of patients around the world. As a global medical technology leader for more than 35 years, we advance science for life by providing a broad range of high performance solutions that address unmet patient needs and reduce the cost of healthcare. For more information, visit www.bostonscientific.com
CONTACTS:
Media:
Laura Aumann
Boston Scientific Corporation
(651) 582-4251 (office)
European Media:
Chiara Ennas
Boston Scientific Corporation
+44 (0) 1442 411618 (office)
Susie Lisa, CFA
Investor Relations
Boston Scientific Corporation
(508) 683-5565 (office)
[i] Medtronic Evera XT VR DVBB2D4 Device Manual page 25.
[ii] Boston Scientific Implantable Cardioverter Defibrillator Physician’s Technical Manual 359050-003 EN US 2014-01 page 30.
[iii] Boston Scientific CRT-D Physician’s Technical Manual 359401-002 EN US 2015-10.
[iv] Quadra AssuraTM Cardiac Resynchronisation Therapy Defibrillator (CRT-D) product manual. https://www.sjm.com/en/professionals/resources-and-reimbursement/technical-resources/cardiac-rhythm-management/cardiac-resynchronization-therapy-crt-devices/crt-defibrillator/quadra-assura-cardiac-resynchronization-therapy-defibrillator-crt-d. Updated August 19, 2013. Accessed February 28, 2017.
[v] Quadra Assura MPTM Cardiac Resynchronisation Therapy Defibrillator (CRT-D) product manual. https://www.sjmglobal.com/en-int/professionals/resources-and-reimbursement/technical-resources/cardiac-rhythm-management/cardiac-resynchronization-therapy-crt-devices/crt-defibrillator/quadra-assura-mp-cardiac-resynchronisation-therapy-defibrillator-crt-d-cd3371-40c-and-cd3371-40qc-with-parylene-coating. Updated December 8, 2015. Accessed February 28, 2017.