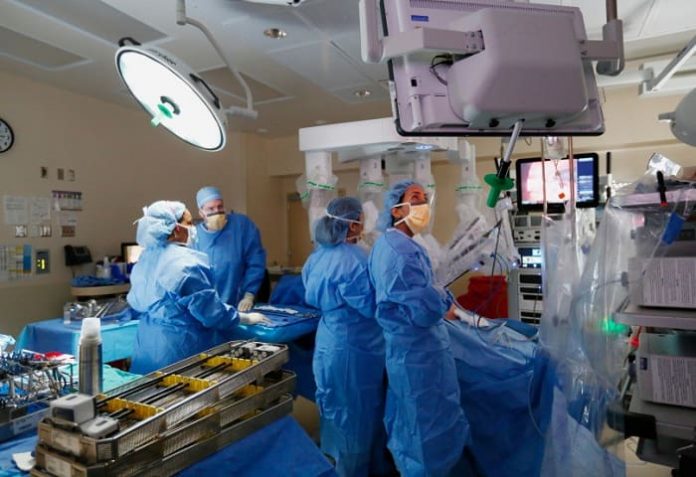
Customer: Intuitive Surgical — Sunnyvale,California USA
Established in 1995, Intuitive Surgical Inc. (Intuitive Surgical) is the global leader in the emerging field of robotic-assisted minimally invasive surgery. Intuitive Surgical’s da Vinci® Surgical System consists of a patient-side cart which includes the robotic arms, a surgeon’s console with 3D HD visualization, and various EndoWrist® instruments used for the specific procedures.
The system translates the surgeon’s natural hand movements at instrument controls on the surgeon’s console into precise, corresponding movements by the robotic arms.
The company has seen double-digit growth since the da Vinci Surgical System was introduced and by 2009, there are over 1,200 systems in hospitals and academic institutions worldwide. The corporate headquarters is in Sunnyvale, California and over 1100 employees and annual sales of over $600 million (USD). Intuitive Surgical was the MD&DI Manufacturer of the year in 2007.
Company history
1995 — Intuitive Surgical founded by Rob Young and Dr. Fred Moll
1997 — Went public, secured main round of funding, and started
development of da Vinci Surgical System
1999 — 1st Generation da Vinci Surgical System receives CE Marking for Europe through UL Notified Body (NB) partnership
2000 — FDA Clearance granted on da Vinci Surgical System
2003 — 1st major upgrade — Added optional 4th arm
2006 — Released da Vinci® STM model — Major redesign of patient side cart, manipulators, and instruments
2009 — Released da Vinci® STM — Second major redesign added Enhanced 3D HD resolution, updated and simplified user interface, upgradeable architecture and compatibility with existing OR suite technology, optional dual console and new ergonomic settings on
the surgeon console.
Why Intuitive Surgical chooses UL
• State-of-the-Art certification knowledge
• Consistent interpretations
• CB Reports accepted worldwide
• High Integrity Certification Marks
• Prestige associated with certification by UL and the UL Mark
New design highlights need for specialized compliance knowledge
Since its inception, Intuitive Surgical Inc. (ISI) has chosen UL as its certification provider. As is typical in the industry, ISI often brought UL in to provide the certification assessment after the product design was complete. When quick questions came up early in development phase, the ISI team would contact the local UL engineering office to see if they could get a quick response. And when those questions would require significant engineering effort, the ISI team would open a preliminary review project.
Opening a formal preliminary review project for each design question often presented challenges because typically, the product was not in a final state of design and may not even have had a formal name. Overall, the established way Intuitive Surgical received certification information did not meet their business needs.
For a major new product design project like the da Vinci Si Surgical System, Mike Yramategui, Principal Regulatory Engineer, knew he needed certification input early in the design process and the availability of UL expertise for consistent interpretation of the requirements of UL / IEC 60601-1.
Working with his UL Account Executive, UL and Mr. Yramategui agreed on a custom contract covering the anticipated scope of work over the new product development timeline. This contract’s scope of services was divided into line items that included a provision for a Dedicated Customer Resource who would hold ‘office hours’ at the Intuitive Surgical facility at regular intervals throughout the year to answer questions related to standards interpretations at ongoing intervals throughout the design process. Due to the innovative nature of the design, the contract also designated a number of additional hours for engineering time to be conducted outside of office hours.
Dedicated customer resource — process
The UL engineer assigned as the Dedicated Customer Resource for Intuitive Surgical Inc. was Staff Engineer, Denise Klinker. The Dedicated Customer Resource provides ongoing service by allocating time for the UL engineer to discuss the standard requirements and their intent. Initially, the Dedicated Customer Resource was introduced to the product and technology and held office hours at the ISI location where the engineers could approach her with questions on standards interpretations, no matter how far along in the process. By getting involved at the beginning of the project, it allowed ISI to make decisions early enough so as not to impact the schedule and design. “By bringing UL in early design stage of product development, this meant they were incorporating safety requirements as design inputs, rather than potential redesign late in the process,” said Denise Klinker.
Having this UL resource was very helpful in supporting Mr.Yramategui during the early design phases. “UL may think we brought them in too early with preliminary ideas. From a customer standpoint, the fact that UL looked at some of those
ideas early and said ’thumbs down’, was very helpful. Internally, being a compliance person, I had cautioned the group that I didn’t think ‘this’ or ‘that’ was going to be acceptable, but it was early enough to say, ‘let’s bring in the experts’ who are going to make the call. UL came in and said, “I don’t see how you can meet this specific clause (or clauses), but if you can provide a risk based rationale on why it achieves an equivalent level of safety we may be able to accept it.’ They were able to help frame it in a way that the engineers could understand why it didn’t comply and, of course then it had the weight of being the UL interpretation rather than the internal person’s interpretation.”
ISI’s design effort involved a core of 5-6 engineering managers, each leading a team of engineers focused on their part of the project. The Dedicated Customer Resource was available to answer the standards questions these engineers had throughout the design process. “It was extremely helpful to be able to send out notices that we have someone coming from UL,which let our staff know that UL is here for any questions on the standards and how they apply to each design area, whether it be mechanical, electrical, thermal, or whatever. This provided a lot of awareness on the project team, and emphasized that the requirements are not just (standard) numbers that the design will automatically meet but it forces them to say “oh what is that?’. It also gave them a convenient way to get information after they realized ‘I do have a question’ on safety factors, or on temperature limits. A lot of the questions may be easy but it offloads me from having to answer hundreds of emails and allows the principals to discuss the issues face to face. ”
When the Intuitive Surgical design team was ready to submit their da Vinci Si Surgical System to UL for certification, Ms.Klinker retained her office hours, and worked with two UL project engineers who were assigned to the project. Two other UL project engineers were assigned to the specific sub-systems and Ms. Klinker’s function as the Dedicated Customer Resource continued as project manager maintaining a consistent level of communication between ISI and UL.
We shared a common goal and the goal was to get it done right and get it done on-time. There’s a secondary aspect … it’s not just schedule. By having that dialogue early, the UL project engineer is a little more comfortable in asking “how does that comply?” because it’s not like at the very end where they don’t want to say anything until it is run through the review chain.
By being able to voice their concerns early, on many occasions we might say ‘oh we can change that, because it’s early in the design phase’. This kind of relationship and communication is important for a safer and better design as well as meeting schedule.”
Collaboration facilitates technical considerations
As expected, during the course of the year, there were some things the UL Dedicated Customer Resource could not answer on her own and she had to work with Intuitive Surgical to understand the situation, and then utilize UL’s network of experts to render an interpretation. This included the PDE, other reviewers, UL’s Software experts, and the project engineers.
In one case, working collaboratively with UL, ISI had to do a complete risk assessment to demonstrate that they met the requirements. This documentation had to go to the UL PDE for review. In a less collaborative scenario, this would have caused an unexpected delay. Being engaged early in the process helped eliminate those issues from negatively impacting the schedule and was a positive experience for the UL engineering team as well. “This process allowed UL to start an early dialogue with the PDE on several constructions
that were not meeting the requirements of the standard,but Intuitive Surgical wanted to incorporate in their design by demonstrating equivalency,” said Ms. Klinker. “Based on discussions with the PDE, we were able to provide preliminary comments and concerns that Intuitive Surgical needed to address in their risk analysis. This gave Intuitive Surgical the ‘head’s up’ on what to expect during the formal investigation if they didn’t want to change their design.”
Proactive review allows for engineering elegant solutions (LEAN Principles)
The proactive review component of the Dedicated Customer Resource office hours allows interpretation questions to be answered during the entire design process. By eliminating redesign and rework for compliance issues, it saves the company time and helps to develop a predictable schedule.
“In the end, it results in better design solutions for compliance because if you find out late in the process, you are going to Band-aid it. If you find out early in the process, you will engineer an elegant solution around it.” As with the LEAN Engineering Principles, this translates into a business benefit to the bottom line.
“The reality of schedules is that they change, they move. This service certainly reduces product and schedule risk,” said Yramategui. “I can’t put a number on that but, as with any development project, your compliance and regulatory activities are dependent on outside agencies so they are somewhat
outside your control, and this has been able to minimize that exposure. This process allows any sort of potential problem to be seen sooner rather than later.”
Complex standard requirements drives need for proactive review
In Mr. Yramategui’s opinion, proactive review and the Dedicated Customer Resource would be a good service option for even less-complex devices. “The standard is sufficiently complex that any new product, even if it is fairly simple would benefit from this model. In the old model, we would try to submit for preliminary construction review early in the design process, but this (DCR service) was even better than that because to get to prelim, the bar is pretty high — you need draft labels, prototypes, and sometimes, that may not occur until it’s too late,or you have made a lot of design decisions.”
Where proactive review may not work
This approach may not necessarily be valuable for design changes where there are typically no interpretation questions, minor construction changes or where the requirements are applied in more of a prescriptive fashion. However, if there is a lack of evidence or precedent that the product revisions will meet requirements, it may still be worthwhile to explore preliminary review or testing prior to final sourcing of materials and parts.
Why Intuitive Surgical chose a dedicated customer resource
• The availability of consistent interpretation among multiple design managers working concurrently on separate parts of the device
• Predictable availability of a compliance engineer to answer interpretation questions
• Dedicated resource available to allocate work
• UL Engineers with expertise in medical device
• Ability to adjust the schedule through communication and sharing of information
• Eliminates the need to open several projects for multiple preliminary reviews to cover all areas of the device
About Intuitive Surgical, Inc.
Intuitive Surgical, Inc. headquartered in Sunnyvale, California, is a global technology leader in robotic-assisted, minimally invasive surgery. Intuitive Surgical develops, manufactures, and markets robotic technologies designed to improve clinical outcomes and help patients return more quickly to active and productive lives. The Company’s mission is to extend the benefits of minimally invasive surgery to the broadest possible base of patients. Intuitive Surgical – Taking surgery beyond the limits of the human hand™.