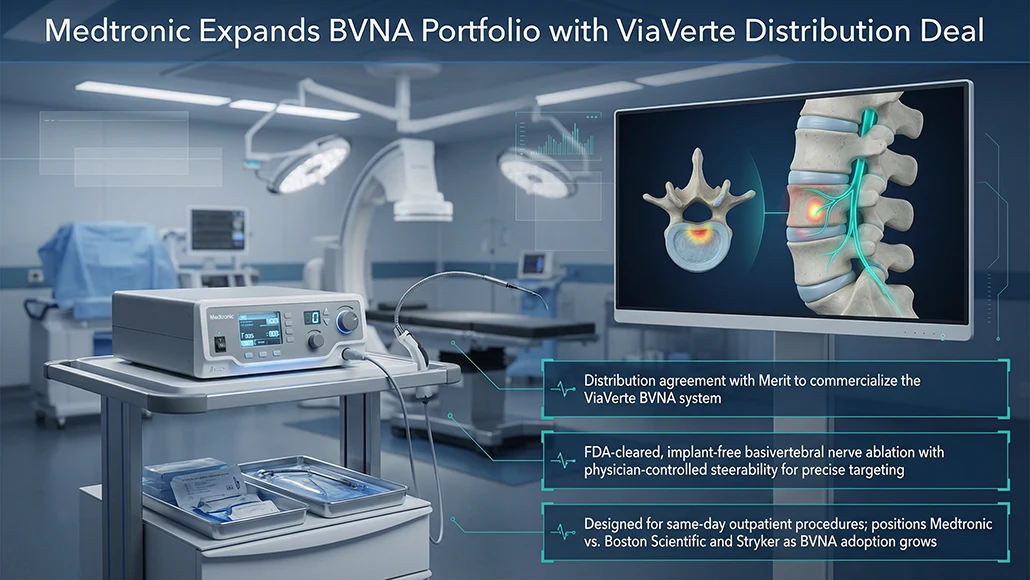
Medtronic has entered into a distribution agreement with Merit Medical Systems to commercialize its ViaVerte system, marking a strategic expansion in the nerve ablation segment. The Medtronic ViaVerte deal brings into focus an FDA-cleared, minimally invasive, implant-free basivertebral nerve ablation (BVNA) system designed to treat chronic vertebrogenic lower back pain. According to Medtronic, the platform stands out as the first and only BVNA system equipped with a physician-controlled steerable mechanism, enabling precise targeting of the basivertebral nerve. The ViaVerte system is intended for same-day outpatient procedures, aligning with broader industry trends toward less invasive pain management solutions.
The Medtronic ViaVerte deal positions the company more directly against competitors such as Boston Scientific, which offers the Intracept BVNA system following its 2023 acquisition of Relievant Medsystems, and Stryker, which received FDA clearance for its own BVNA system in May 2025. Medtronic indicated that the agreement builds on its series of strategic initiatives aimed at strengthening core franchises. It also deepens its collaboration with Merit Medical, which currently supplies the Kyphon KyphoFlex unipedicular balloon catheter. The companies stated that the ViaVerte BVNA system is expected to become available later this year.
“For 50 years, we have advanced how chronic pain is treated,” said Paolo Di Vincenzo, president, Medtronic Neuromodulation, which is part of the Medtronic Neuroscience Portfolio. “Adding ViaVerte basivertebral nerve ablation expands our world-leading pain interventions portfolio and gives patients and their physicians another meaningful option for lasting relief.”
“We are thrilled to expand our ongoing business relationship with Medtronic by providing Merit’s proprietary articulating technology. We believe ViaVerte represents a significant advancement in the BVNA market.”
“This agreement underscores our mutual dedication to providing innovative therapies to support physicians and their patients.”
BTIG analysts Sam Eiber, Marie Thibault and Alexandra Pang maintained a “Buy” rating for Merit following the announcement. They highlighted that limited details were disclosed about ViaVerte beyond its physician-controlled steerable mechanism. While describing the BVNA segment as a “relatively small market” estimated at around $200 million, the analysts pointed to its rapid growth, supported by activity from Boston Scientific and Stryker. “We are pleased to see Merit enter this fast-growing and under penetrated market and think Medtronic makes sense as a partner given its Neuromodulation portfolio,” the analysts wrote. “It could also serve as one lever to help the OEM business get back on more stable footing.”