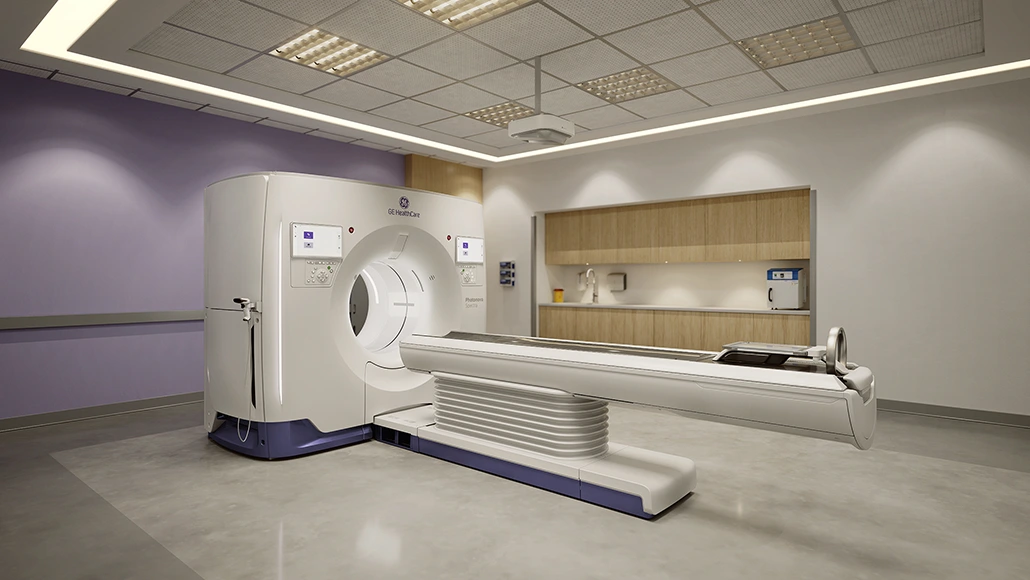
GE HealthCare has secured FDA clearance for its Photonova Spectra solution, marking a significant step in the advancement of photon-counting computed tomography. The Chicago-based company confirmed that the system received FDA 510(k) clearance, positioning Photonova Spectra as a flexible PCCT platform designed to address a wide range of clinical requirements. Built on GE HealthCare’s proprietary Deep Silicon detector technology, the system introduces multiple configuration options aimed at supporting diverse imaging environments while enhancing diagnostic precision.
The Photonova Spectra platform is engineered to deliver broad coverage alongside ultra-high definition (UHD) spatial and spectral imaging. According to GE HealthCare, the system enables faster acquisition speeds and supports detailed visualization of subtle tissue variations, small lesions and vascular structures. Unlike conventional CT systems that convert X-rays into light before measurement, photon-counting CT directly detects individual X-ray photons and measures their energy. This approach allows for improved spatial and spectral resolution, as well as enhanced tissue characterization. The company also highlighted that its Deep Silicon detector material strengthens spectral imaging performance, particularly in lesion characterization and treatment monitoring. GE HealthCare submitted Photonova Spectra for FDA clearance in November 2025, incorporating advanced AI applications into the platform.
As clinicians across the United States face rising volumes and increasing diagnostic complexity, technology must do more than capture images it must simplify decision-making and strengthen performance across the enterprise, said Catherine Estrampes, President & CEO, U.S. and Canada, GE HealthCare. Photonova Spectra is designed to deliver rich clinical insights in every scan and help alleviate cognitive burden for care teams. With the U.S. 510(k) clearance, we are proud to now bring this innovation to U.S. healthcare systems and the patients they serve.
Further technical capabilities of Photonova Spectra include 8-bin energy resolution enabled by Deep Silicon, supporting advanced material separation and characterization. This allows clinicians to distinguish materials such as iodine, calcium and fat with greater clarity. The system’s wide detector coverage and rapid rotation speed of 0.23 seconds contribute to fast acquisition and motion-free imaging. Additionally, Photonova Spectra captures both 8-bin spectral and ultra-high definition spatial data simultaneously without requiring specialized protocols, ensuring spectral data is available in every exam.
GE HealthCare noted that the system has applications across neurology, oncology, musculoskeletal imaging, thoracic imaging and cardiology. It supports visualization of fine brain structures, characterization of lesions, detection of small fractures and bone marrow edema, and advanced cardiac and chest imaging. The platform also leverages Nvidia computing technology, enabling it to process up to 50 times more data than conventional CT systems. Ongoing collaborations with UW-Madison and Stanford Medicine are focused on exploring new clinical applications and imaging protocols. With regulatory approval in place, GE HealthCare is now preparing for commercial availability of Photonova Spectra in the U.S.
Photonova Spectra reflects years of intentional design and close collaboration with clinicians, researchers and collaborators across the globe, adds Jean-Luc Procaccini, president & CEO, Molecular Imaging and Computed Tomography, GE HealthCare. From the earliest stages to today, we remain focused on building a system that addresses the practical realities of clinical practice while opening pathways for scientific advancement. The result is a photon-counting platform engineered for the needs of today’s care teams, as well as the imaging challenges and research opportunities that will shape the future of CT.