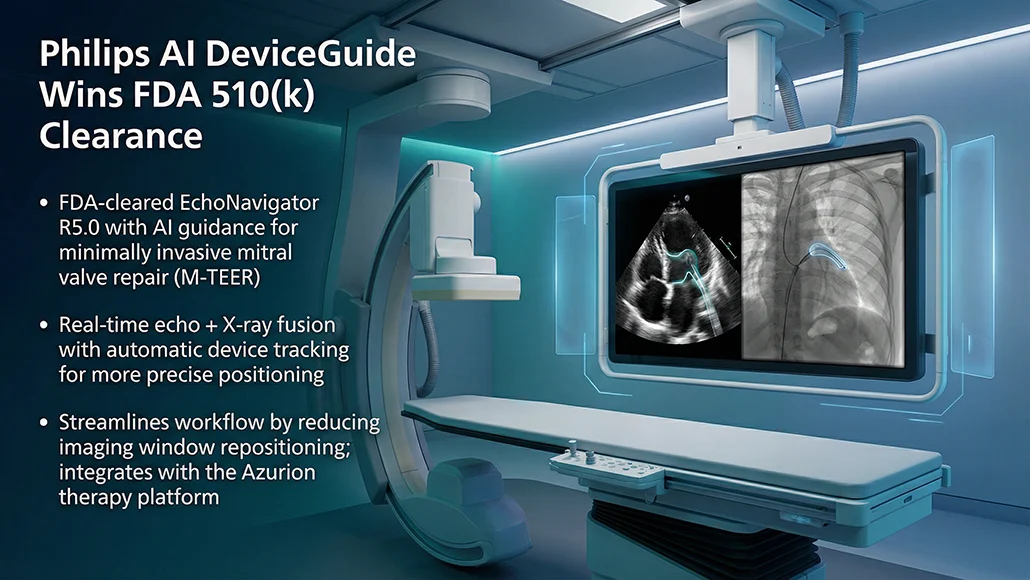
Regulatory clearance has been secured by Philips from the U.S. Food and Drug Administration for EchoNavigator R5.0 with DeviceGuide, an artificial intelligence-based software developed to assist physicians during minimally invasive mitral valve repair procedures. Designed to address the technical complexity of these interventions, the platform delivers real-time visualization and procedural guidance. The advancement reinforces the growing role of AI DeviceGuide in supporting precision-led interventional cardiology.
The development of DeviceGuide took place in collaboration with Edwards Lifesciences, combining Philips’ imaging and AI capabilities with Edwards’ expertise in structural heart therapies. The software is tailored to enhance workflow efficiency during mitral transcatheter edge-to-edge repair (M-TEER), which serves as a minimally invasive alternative to open-heart surgery for patients with mitral regurgitation. Through workflow optimization and guided navigation, AI DeviceGuide supports clinicians in executing these demanding procedures with improved consistency.
At its core, the system leverages Philips’ echo-fluoro fusion technology to merge live ultrasound and X-ray imaging into a unified display. Its AI-driven algorithm automatically tracks and visualizes the repair device in real time, enabling more precise positioning during procedures. “The AI software serves as an assistive tool; the physician always remains in control. This isn’t about replacing expertise – it’s about amplifying it,” said Atul Gupta.
Development efforts included collaboration with investigational sites across Europe and the U.S., notably at the Structural Heart and Valve Center at NewYork-Presbyterian/Columbia University Irving Medical Center. The work involved Susheel Kumar Kodali, MD, and Rebecca T. Hahn, MD. “Since AI auto-aligns imaging to the device in real time and continuously informs the interventionalist about the location of the device in space on the imaging screen, it minimizes unnecessary repositioning of the imaging window, streamlines procedural guidance, and may improve the precision of device implantation,” said Dr. Rebecca T. Hahn. The solution integrates with Philips’ Azurion image-guided therapy platform and aligns with its broader connected cardiology strategy.