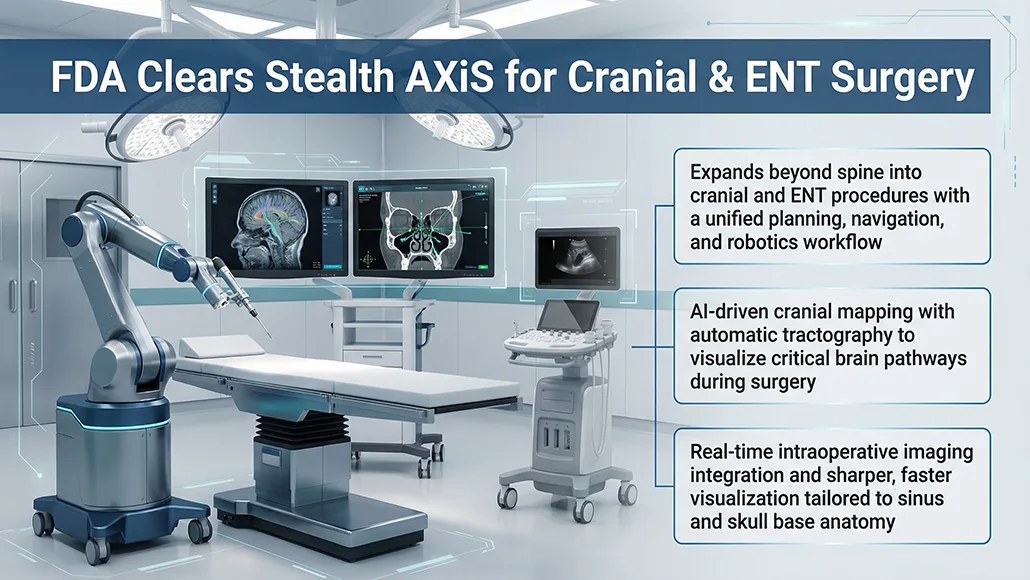
Medtronic’s Stealth Axis FDA clearance marks an expansion of its surgical system into cranial and ear, nose, and throat (ENT) procedures, following regulatory approval from the US Food and Drug Administration. The clearance builds on the system’s earlier authorisation for spine procedures in February 2026, positioning Stealth Axis as a unified platform that combines surgical planning, navigation, and robotics. Designed to deliver precision and real-time intraoperative insight, the system integrates multiple surgical capabilities into a single workflow environment.
At the core of Medtronic’s offering is an artificial intelligence-driven architecture embedded within the Stealth Axis platform. The system includes a floor-mounted robotic arm alongside dedicated viewing consoles, enabling surgeons to access planning and visualisation tools before, during, and after procedures. Within cranial applications, the platform introduces AI-based automatic tractography, allowing for the creation of patient-specific brain maps and enabling visualisation of critical neural pathways tied to essential brain functions. It also incorporates real-time imaging capabilities through integration with GE HealthCare’s bkActiv ultrasound system.
For ENT procedures, the system has been developed around established clinical workflows, providing navigation and advanced visualisation tailored to the complexities of sinus and skull base anatomy. According to Medtronic, the system delivers sharper anatomical detail and faster responsiveness, supporting surgical decision-making in the operating room (OR). The company positions Stealth Axis as the “cornerstone” of its AiBLE ecosystem, which connects imaging, data analytics, and software platforms to streamline surgical workflows across spine, cranial, and ENT procedures.
Michael Carter, senior vice president and president of Medtronic cranial and spinal technologies, commented: “Cranial procedures require an exceptional level of precision, planning, and real-time insight.
“With the Stealth AXiS surgical system, we are bringing together advanced navigation, robotics, and AI-enabled capabilities within the AiBLE smart ecosystem to support surgeons with deeper insights across the surgical workflow.”