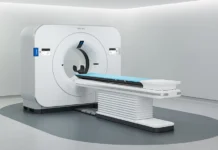
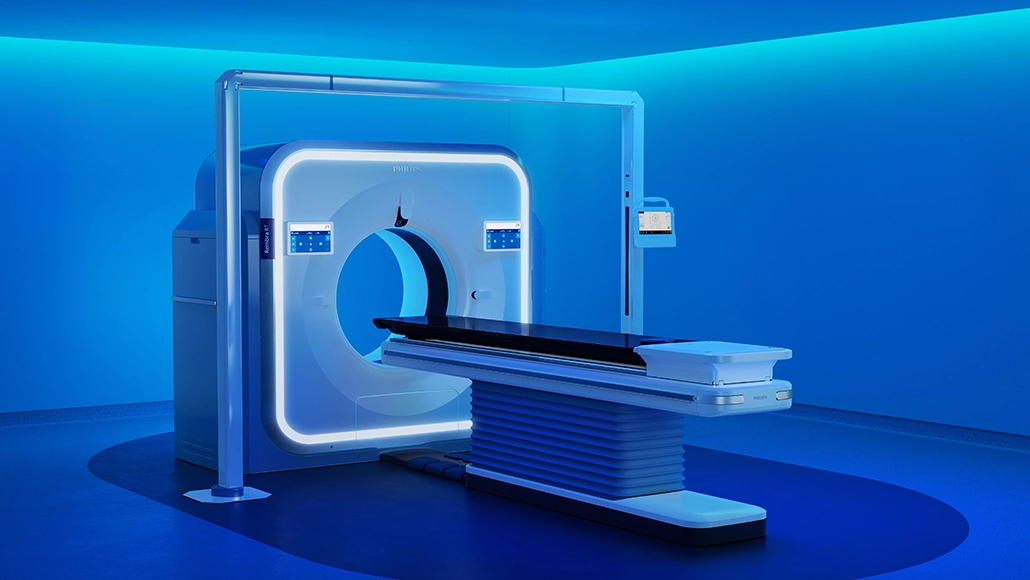
Philips has secured FDA 510(k) clearance for its Rembra platform of scanning systems, marking a regulatory milestone for its latest computed tomography technologies. The clearance covers the Rembra CT, Rembra RT and Areta RT systems, which are positioned to support both diagnostic imaging and radiation therapy planning. The company had introduced the Rembra CT system in March 2026, following earlier debuts of Rembra RT and Areta RT in September 2025, along with a detector-based spectral CT system powered by AI unveiled in December 2025.
The newly cleared Rembra platform is designed to help healthcare providers manage rising demand for imaging services while improving operational efficiency and access to high-quality diagnostics. According to Philips, the platform extends the application of computed tomography across radiology and radiation therapy, enabling more integrated workflows and coordinated patient management. The systems aim to address clinical complexity by supporting faster imaging processes and more streamlined decision-making pathways.
Within the platform, Rembra CT features an 85-cm bore and is engineered for high-throughput environments, allowing up to 270 exams per day across emergency, critical care and interventional settings. Meanwhile, Rembra RT and Areta RT bring similar imaging capabilities into radiation therapy, supporting treatment planning with high-fidelity imaging, an extended field of view and next-generation 4DCT functionality. Collectively, the Rembra platform establishes a unified CT ecosystem designed to operate across the full care continuum.
Dan Xu, business leader of CT at Philips, said:
“As healthcare systems manage increasing demand and complexity, imaging plays a critical role in enabling timely and informed clinical decisions. With the Rembra platform, we are redefining what clinicians can expect from CT, combining speed, scalability, and precision to expand access to high-quality imaging while supporting confident diagnosis and highly accurate treatment planning.”