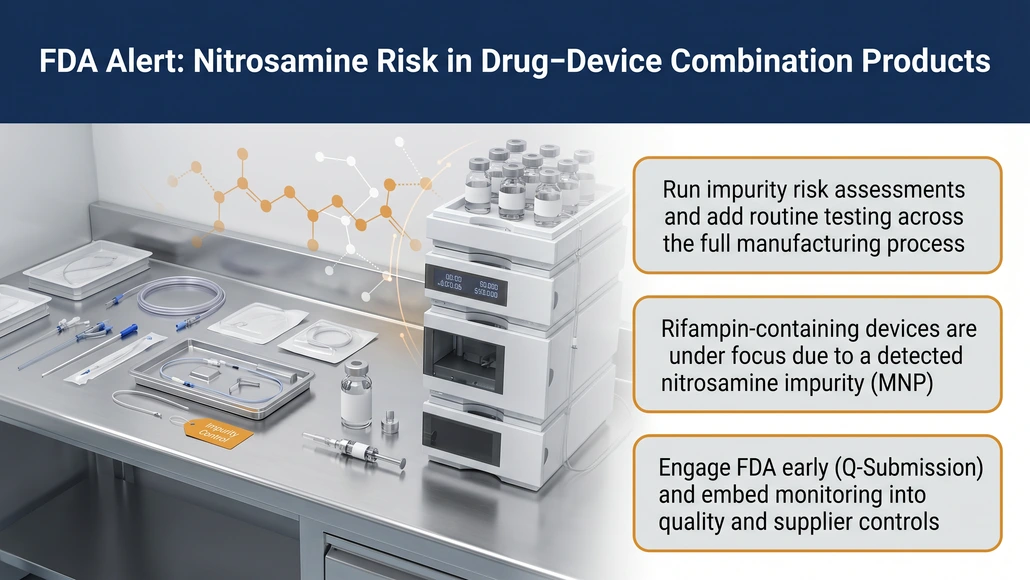
The U.S. Food and Drug Administration (FDA), through its Center for Devices and Radiological Health (CDRH), has issued a formal communication to manufacturers of CDRH-led combination products, highlighting the nitrosamine impurity risk and outlining regulatory expectations for risk identification, mitigation, and ongoing monitoring. The move underscores increasing regulatory scrutiny on drug-device combination products, particularly those incorporating pharmaceutical components such as rifampin.
The agency’s letter provides detailed guidance on manufacturer responsibilities, while reinforcing the need for proactive engagement with regulators to ensure continued product safety and effectiveness.
Regulatory Direction and Manufacturer Responsibilities
The FDA’s communication establishes a clear compliance framework for healthcare manufacturers. It emphasizes that organisations must take full responsibility for identifying potential nitrosamine impurities originating from both pharmaceutical ingredients and manufacturing processes.
Manufacturers are expected to:
- Conduct comprehensive risk assessments as part of biocompatibility evaluations
- Develop and deploy analytical testing methods to detect and control impurities
- Assess patient exposure levels relative to FDA-defined acceptable intake limits
- Investigate all possible contamination pathways, including manufacturing, sterilisation, and storage
The agency also advises manufacturers to consult existing FDA guidance, including standards related to nitrosamine control and biological evaluation frameworks such as ISO 10993-1.
Identified Risks in Drug-Device Combination Products
CDRH has identified the presence of 1-methyl-4-nitrosopiperazine (MNP) a nitrosamine impurity in certain combination products containing the antibiotic rifampin. Nitrosamines are classified as probable carcinogens, and prolonged exposure above acceptable thresholds may elevate cancer risk.
While no specific products or manufacturers have been named, the agency highlighted multiple categories of devices where rifampin is commonly used:
- Antimicrobial-coated catheters, including central venous and ventricular variants
- Cardiac implant accessories such as antibacterial envelopes used with implantable cardioverter defibrillators (ICDs)
- Neurostimulation and neuromodulation implants
- Prosthetic devices incorporating antibiotic surface treatments
Ongoing Investigation and Regulatory Engagement
The FDA confirmed that investigations into the root causes and scope of nitrosamine formation are ongoing. At present, the agency has not identified any adverse events linked to these impurities in CDRH-regulated products. However, the absence of reported cases has not reduced the urgency of regulatory action.
CDRH is working closely with manufacturers to:
- Determine the mechanisms behind nitrosamine formation
- Evaluate the applicability of interim or alternative exposure limits
- Standardise risk assessment methodologies across product categories
Manufacturers identifying potential risks are advised to engage with the FDA via the Q-Submission process, enabling early regulatory feedback and alignment on mitigation strategies.
Operational and Compliance Implications for Healthcare Management
From a healthcare management perspective, the FDA’s directive introduces heightened compliance expectations across product lifecycle management. Organisations must integrate impurity risk assessment into broader quality systems, particularly in areas involving supply chain oversight, raw material sourcing, and manufacturing validation.
As observed by HHM Global, this development signals a shift toward more integrated regulatory oversight of combination products, where pharmaceutical and device standards increasingly converge. HHM Global notes that manufacturers will need to strengthen cross-functional coordination between regulatory affairs, quality assurance, and production teams to manage emerging risks effectively.
The emphasis on nitrosamine impurity risk also reinforces the importance of data-driven monitoring frameworks, ensuring that potential contamination sources are identified early and mitigated before reaching clinical use.
Strategic Industry Relevance
The FDA’s action reflects a broader regulatory trend toward tighter control of impurities in complex healthcare products. For manufacturers, this translates into increased investment in analytical capabilities, enhanced supplier collaboration, and more rigorous validation protocols.
In practical terms, HHM Global highlights that this move could reshape compliance strategies for combination product manufacturers, particularly those relying on antibiotic-impregnated devices. The regulatory focus on impurity pathways from raw materials to finished products positions nitrosamine risk management as a critical component of long-term product safety governance.