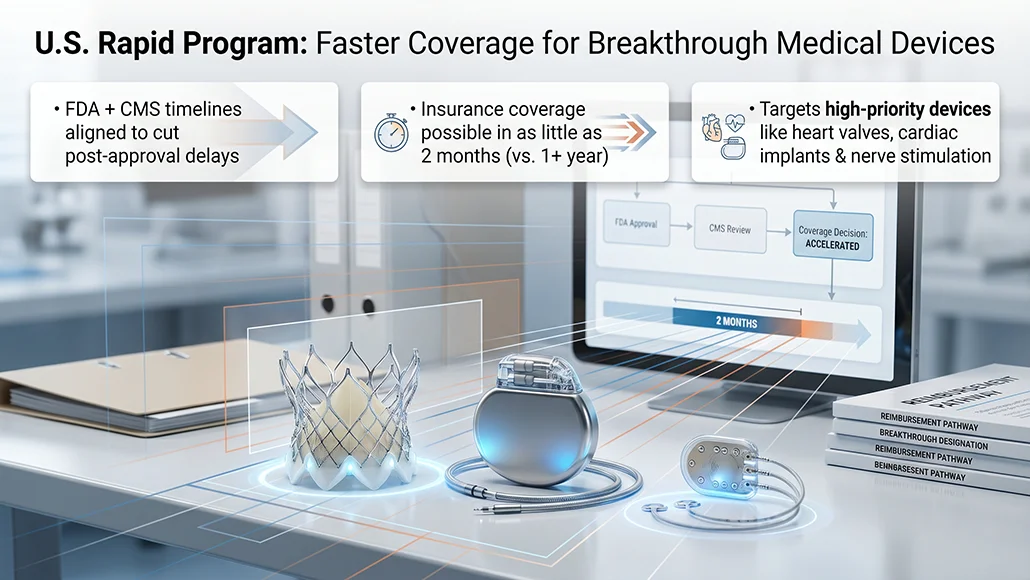
A new federal initiative in the United States is set to significantly shorten the timeline for securing insurance access to advanced medical technologies, addressing a long-standing bottleneck in patient availability and commercial rollout. The program introduces a coordinated approach between regulatory approval and reimbursement decisions, allowing certain high-priority innovations to move more swiftly from development to real-world use. Under this framework, a Breakthrough Medical Device could receive insurance coverage in as little as two months substantially faster than the traditional process, which often extends beyond a year.
The initiative, jointly introduced by the Centers for Medicare and Medicaid Services and the U.S. Food and Drug Administration, is formally known as the Regulatory Alignment for Predictable and Immediate Device, or Rapid. It is designed for technologies that already qualify for expedited regulatory review due to their potential to address unmet medical needs. By synchronising FDA evaluation timelines with CMS coverage considerations, the program integrates reimbursement planning earlier in the product lifecycle, reducing delays that typically arise after regulatory clearance.
“The American people deserve timely access to meaningful treatments without red tape or high costs,” FDA Commissioner Marty Makary said in a statement.
Devices expected to fall within the scope of the program include artificial heart valves, cardiac rhythm management technologies, and nerve-stimulation implants used in disease treatment. According to CMS chief policy and regulatory officer John Brooks, approximately 40 devices currently meet eligibility criteria, with an additional 20 potentially qualifying. These products are often developed by leading medical technology companies such as Medtronic Plc, Boston Scientific Corp., and Abbott Laboratories, reflecting the high level of innovation targeted by the pathway.
- The program aligns FDA approval timelines with CMS reimbursement decisions
- Coverage timelines may shrink from over a year to as little as two months
- Around 40 devices are immediately eligible, with 20 more under consideration
- Focus areas include cardiac devices, implants, and nerve stimulation technologies
For device manufacturers, delays in securing reimbursement have long represented a critical commercial and operational challenge, frequently extending development cycles and limiting market entry. The fragmented nature of coverage where access varies by region has further complicated adoption. Central to this issue is the National Coverage Determination process, which defines whether Medicare and Medicaid will reimburse specific technologies. Although private insurers are not formally bound by these decisions, they often align with federal determinations, amplifying their industry-wide impact. The new framework aims to reduce these inefficiencies and provide a more predictable pathway for each Breakthrough Medical Device entering the healthcare system.