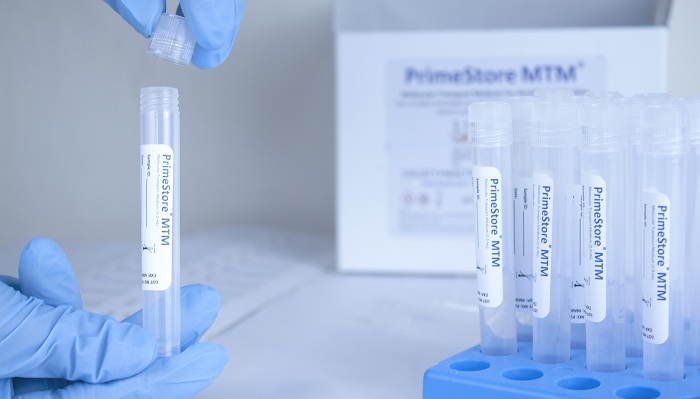
EKF Diagnostics, the global in vitro diagnostics company, announces it has secured new contracts for the manufacture and supply of a novel, patented sample collection device which allows COVID-19 samples to be rapidly inactivated in the collection tube, avoiding contamination and preserving RNA without need for refrigeration. To support the increase in COVID-19 testing globally, a safe and easy sample collection and transport mechanism is essential, this is provided by the PrimeStore® MTM sample collection device.
EKF is a contract manufacturer and supplier for the PrimeStore MTM, which is an FDA-cleared and CE-marked microbial nucleic acid storage and stabilization device, owned and created by Longhorn Vaccines and Diagnostics LLC (Bethesda, Maryland, US). PrimeStore MTM deactivates viruses, bacteria, fungi and mycobacterium tuberculosis allowing safe sample handling and transport, and eliminating the risk of infection during transport and in laboratories. In addition to removing the need for cold chain storage of samples, RNA and DNA are perfectly preserved ready for safe testing immediately on arrival at a laboratory and without need for containment. COVID-19 has led to a significant increase in global demand for this unique device, which is the only FDA Class II cleared device for microbial nucleic acid storage and stabilization (RNA and DNA) available for the safe transportation of samples which may contain viruses.
In addition to more than doubling its current manufacturing capacity of this device in the US on behalf of Longhorn, EKF has now rapidly repurposed its European manufacturing facilities in Cardiff, UK, and soon Germany. This substantial increase in manufacturing capacity is to meet not only new US orders, but also new European supply contracts for PrimeStore MTM; including an agreement with Source BioScience UK Ltd to be used for its growing COVID-19 testing service.
Source BioScience provides ISO:15189 laboratory testing services for the NHS and private healthcare. It is working with a number of businesses to provide them with COVID-19 testing services for their staff to enable them to bring back those who may be self-isolating due to concerns over infection, or that of a family member. Being able to use the PrimeStore MTM transport media allows a significant benefit of removing the cold chain transportation that is a necessary protocol for other transport media.
“We are delighted to supply Longhorn’s PrimeStore MTM collection kits to Source BioScience, a well-established provider of testing services to the NHS. These collection kits will ensure that COVID-19 samples can be handled and transported without expensive refrigeration or fear of contamination,” said Julian Baines, CEO of EKF Diagnostics. “This is a key part of the testing supply chain which can underpin the safe and rapid increase of testing capacity. In addition to the US, we will continue to ramp up our production capacity here in the UK and in Europe to meet increasing demand and support efforts to limit the spread of Coronavirus globally.”
Jay LeCoque, Chairman and CEO of Source BioScience, added, “We are very encouraged with this partnership and significant development from EKF, which allows the removal of cold chain transportation, making logistics of samples less expensive and more available to the many hospitals and businesses needing to regularly test key workers and staff. This dramatically expands the reach of COVID-19 testing services to a larger population and will support a faster economic recovery.”
PrimeStore MTM was invented and patented by Longhorn Vaccines and Diagnostics LLC in 2006 in preparation for a worldwide pandemic and has already been used in testing for many infectious diseases and high consequence pathogens including influenza, RSV, TB, HIV and coronavirus. As it inactivates pathogens rapidly and stabilises the RNA for up to four weeks with no requirement for cold storage, this makes the logistics of sample collection less expensive and allows testing to be more readily available to institutions and businesses that need to test staff regularly. Furthermore, in the case of COVID-19, by inactivating the virus, testing can now take place outside of containment facilities opening up capacity for more testing laboratories.
About EKF Diagnostics
EKF Diagnostics Holdings plc, which includes the EKF Diagnostics, Stanbio Laboratory and DiaSpect brands, specializes in the development, production and worldwide distribution of point-of-care analyzers for use in the detection and management of diabetes and anemia, and also in sports and maternal medicine. EKF products are sold in more than 100 countries around the globe, through a network of specialist distributors.
Point-of-care diagnostics: EKF Diagnostics designs and manufactures world-class diagnostic devices, as well as distributing rapid test kits for infectious diseases and pregnancy. The EKF analyzer range is used widely in GP surgeries, pharmacies, blood banks, sports clinics, hospitals and laboratories for glucose, lactate, hemoglobin, hematocrit and HbA1c measurement.
Central laboratory: EKF, through its wholly owned subsidiary, Stanbio Laboratory (Boerne, Texas, USA), manufactures a comprehensive range of clinical chemistry reagents, as well as associated analyzers. In addition, EKF Life Sciences (Elkhart, Indiana, USA) manufactures enzymes used in reagent development and also provides contract fermentation facilities.