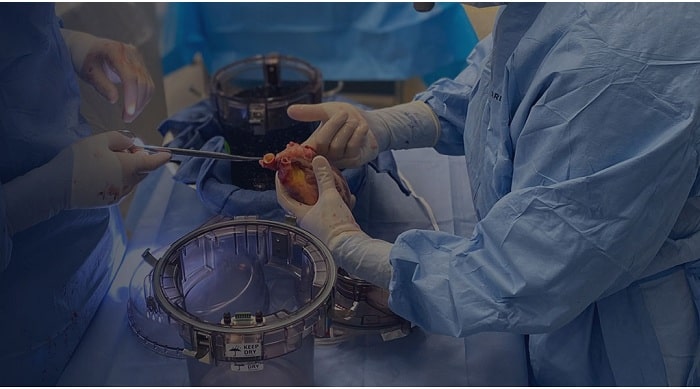
Paragonix Technologies, Inc. is announcing a major milestone of 1,000 preserved donor heart transports since it began clinical use in 2018, with its flagship product, the Paragonix SherpaPak® Cardiac Transport System (CTS). The Paragonix SherpaPak CTS is the leading FDA cleared and CE marked medical device for heart preservation and transport. Four of the top five largest heart transplant programs in the US and 45 programs globally now trust the Paragonix SherpaPak System to preserve and transport critical donor hearts to their recipient donors.
The Paragonix SherpaPak CTS protects donor hearts with its rigid, sterile container and maintains a temperature between 4-8ºC, a temperature range recommended by the International Society of Heart and Lung Transplantation as an optional donor heart preservation condition1. The Paragonix SherpaPak CTS replaces the consumer-grade cooler and ice system that has been used historically – a method that was long overdue for innovation. Wireless connection to a simple app gives organ environment tracking and real-time updates to the transplant team, providing the transplant teams with information critical to this complicated surgery.
“Reaching this milestone of 1,000 donor hearts preserved is a tremendous accomplishment for us,” said Dr. Lisa Anderson, CEO of Paragonix. “This is a huge breakthrough for the medical community and, with surgeons using our technology to travel from surgical center to surgical center, we are allowing donor hearts to safely travel farther and longer while also ensuring they arrive in a viable condition for transplant.”
Nationally, clinical studies and publicly available UNOS data have shown an increase in distance traveled and ischemic time since changes were made to UNOS Allocation Policy in 2018.2,3 Recently, two leading transplant centers have published clinical studies, not only highlighting this trend, but suggesting that this increase in ischemic time and has been correlated to an increase in Primary Graft Dysfunction (PGD) and potential injury to the donor organs.2,3 A recent multi-center study published in the ASAIO Journal cited a direct correlation between increase in ischemic time and PGD rates stating that “Each 10 minute increase in ischemic time was associated with 5% increased odds of PGD.”4
A new case report out of Stanford, published in Transplant Proceedings, found that the temperature-controlled SherpaPak Cadiac Transport System may help with challenges related to increased ischemic times.5 Additionally, the first report of the GUARDIAN-HEART Registry, a global clinical registry looking at the impact of organ preservation on clinical outcomes, found that in a comparative study looking at over 200 clinical cases, a 54% reduction in Severe PGD (p=0.071) for cases using the Paragonix SherpaPak compared to clinical cases using ice storage. 6 Even more significantly, during the same comparative study which demonstrated this reduction in Severe PGD, the clinical cases utilizing the Paragonix SherpaPak system had on average a 13% longer ischemic time (p=0.0003) than the clinical cases using ice storage.6
Professor Andreas Zuckermann, Director of Cardiac Transplantation at the Vienna Cardiac Transplant Group at the Medical University of Vienna and one of the earliest adopters of this technology, commented on this milestone, “Knowing that over 1,000 hearts have been transported within the Paragonix SherpaPak system in such a short period of time is a gratifying testament to the collaboration between science and industry to find new ways to protect donor hearts. Organ donation is such a precious gift, and these advancements help ensure that we as clinicians can make the most of their impact on a transplant recipient’s life.” Dr. Zuckermann, is also a co-principal investigator of the GUARDIAN study investigating the impacts of the Paragonix device. “We now have studied more than 500 patients from the GUARDIAN registry and the data continues to show increasingly promising clinical benefit of Paragonix SherpaPak system on patient outcomes. As more and more cases are added to this important registry, our team is growing more and more confident that this system can limit risk and help reduce transplant complications.”
Paragonix’s recently announced OPTION program, a unique network of holding sites across the US, ensures that the choice for advanced donor organ preservation with the Paragonix systems is never too far away. This inventory of responsive depots will ensure transplant surgeons have access to Paragonix’s preservation technologies at any given time.
About Paragonix Technologies
Paragonix Technologies markets organ transportation devices that safeguard organs during the journey between donor and recipient patients. Our devices incorporate clinically proven and medically trusted cold preservation techniques in a novel suspension system to provide unprecedented physical and thermal protection. Paragonix SherpaPak® CTS is the only commercially available FDA cleared and CE marked transport device for heart transportation.