AliveCor, the leader in AI based, personal ECG technology, and provider of enterprise cardiology solutions; and Huami Corporation, a global leader in biometric, data-driven wearables, announced a multi-faceted global strategic alliance that combines the technologies of each company to deliver a new generation of medical functionality to wearable devices.
“AliveCor’s Intelligent, medical grade, heart care platform will be a key component of Huami smart watches and heart trackers going forward. Our new partnership is critical to building these as well as entirely new medical grade services,” said Wang Huang, CEO of Huami.
“AliveCor’s agreement with Huami achieves our corporate goal of delivering AliveCor’s AI driven subscription services and solutions to enterprise customers and consumers worldwide at affordable price points. AliveCor technology allows partners to easily deliver clinically validated, peer-reviewed medical features to their own customers, at scale,” said AliveCor CEO Priya Abani.
The agreement announced today encompasses the licensing of AliveCor technology and an agreement to work together to bring new devices and services to market. The companies expect that the alliance will deliver entirely new ECG form factors to global markets in 2020.
About Huami Corporation
Huami is a biometric and activity data-driven company with significant expertise in smart wearable technology. Since its inception in 2013, Huami has quickly established its global market leadership and recognition by shipping millions of units of smart wearable devices. In 2018, Huami shipped 27.5 million units of smart wearable devices. Huami has one of the largest biometric and activity databases in the global smart wearables industry. Huami’s mobile apps work hand in hand with its smart wearable devices and provide users with a comprehensive view and analysis of their biometric and activity data.
About AliveCor
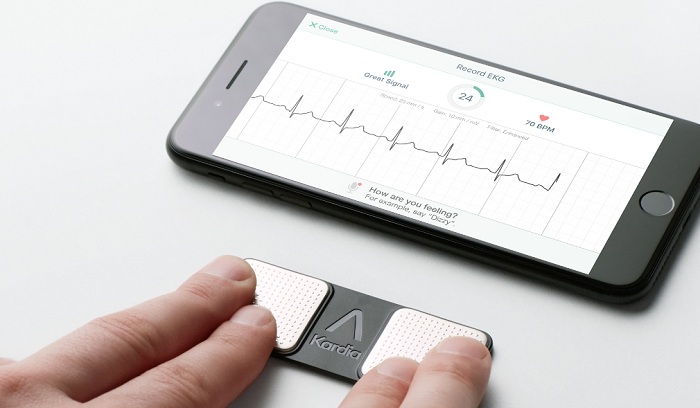
AliveCor, Inc. is transforming cardiological care using deep learning. The FDA-cleared KardiaMobile is the most clinically validated personal ECG solution in the world. KardiaMobile provides instant detection of Atrial Fibrillation, Bradycardia, Tachycardia, and Normal rhythm in an ECG. Kardia is the first AI enabled platform to aid patients and clinicians in the early detection of atrial fibrillation, the most common arrhythmia and one associated with a five times greater risk of stroke. AliveCor’s KardiaPro enterprise platform allows practitioners and institutions to manage chronic heart conditions simply and profitably using KardiaMobile. AliveCor was named the No.1 artificial intelligence company in Fast Company’s Top 50 Most Innovative Companies. AliveCor is a privately-held company headquartered in Mountain View, Calif. “Consumer” or “Personal” ECGs are ECG devices available for direct sale to consumers.