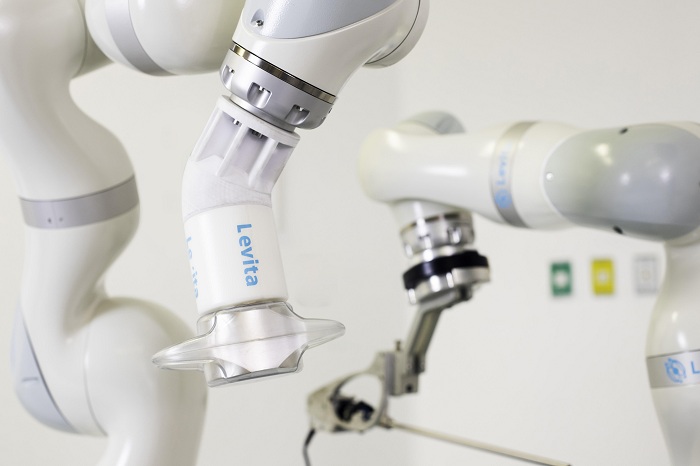
Levita Magnetics announced that it performed the first procedures with its robot-assisted surgical platform. Dr. Ignacio Robles used Menlo Park, Calif.–based Levita’s newest system, the Levita Robotic Platform, in a reduced-incision laparoscopic cholecystectomy (gallbladder removal). The procedure took place at Clínica INDISA in Santiago, Chile, as part of a clinical study of the platform.
According to a news release, Levita designed the new platform to deliver the clinical benefits of the company’s first commercial product, the Levita Magnetic surgical system, which includes less pain, faster recovery and fewer scars. The company’s technology involves a magnet controller outside the body moving tools inside the body.
The company said it intends to use the platform to improve visualization, maintain surgeon control of instruments and increase hospital efficiency with fewer assistive personnel required for the procedure. The design specifically targets high-volume ambulatory or same-day discharge abdominal surgeries.
Robles said the first procedure “went very smoothly” and delivered positive recovery with no complications at 30 days post-surgery. Two additional procedures have been performed since, with Robles touting Levita’s magnetic technology to improve surgeon control and efficiency.
“We are taking magnetic surgery to the next level with this disruptive approach. Our robotic platform is designed to be the first to enable the clinical benefits of a less invasive procedure with fewer incisions while allowing the surgeon full control of the platform and surgical instruments directly beside the patient,” said Levita Magnetics founder & CEO Dr. Alberto Rodriguez-Navarro.
“We believe enabling timely access to needed surgery has a direct impact on the quality of life for patients and may reduce the risk of complications or death due to the underlying disease,” Rodriguez-Navarro said. “Our ultimate goal is to globally increase the ability of surgeons and hospitals to provide more patients access to better surgical procedures within their communities.”
The company is advancing its clinical study and plans to submit the Levita Robotic Platform to the U.S. FDA for clearance in late 2021.