Depending on the health condition of the patient and additionally prescribed drugs and/ or chemotherapy, fenbendazole daily dosage can vary.
Dosage guide:
Note: by taking chemotherapy, additional supplements, natural products, nutraceuticals or pharmaceuticals not related to cancer treatment together with fenbendazole and to decide whether fenbendazole can be taken alone to treat cancer, one should consult with professional, licensed medical consultant who has appropriate attitude and experiance in fields of traditional chemotherapeutic and combinatorial (alternative) cancer treatment and experiance in medication reconciliation to prevent adverse efects when taking several drugs/chemicals together.
Storage of fenbendazole powder, fenbendazole capsules and fenbendazole tablets: below 25 °C and not exposed to sun.
Toxicity, side effects and metabolism of fenbendazole
Toxicity, genotoxicity and carcinogenicity
It is hard to evaluate what certain health and side effects normally or highly dosed fenbendazole may cause for humans, as it is not a medication for people and there is no previous history of human-usage. It is even more difficult to predict the side effects when additional medication is taken together with fenbendazole.
Fenbendazole alone has a very low acute toxicity when consumed orally (measured in rodents). No acute oral exposure limit is determined (it may be that acute toxicity threshold is above 5000mg of material per kilogram of body weight).
Based on human data it appears that doses up to 500 mg per person did not result in any adverse effects. Additionally, single doses up to 2,000 mg per person also caused no adverse effects.
Based on a variety of in vivo and in vitro genotoxicity tests, where European Medicines Agency was involved and partially responsible, it was determined that fenbendazole is not genotoxic.
I evidence for carcinogenicity was found in a 2-year study in mice.
Data about toxicity, genotoxicity and carcinogenicity retrieved from.
Side effects:
Side effects include:
- Vomiting (rare)
- Diarrhea (rare)
- Jaundice (very rare)
- Skin itching (very rare)
- Liver injury (extremelly rare)
Metabolism
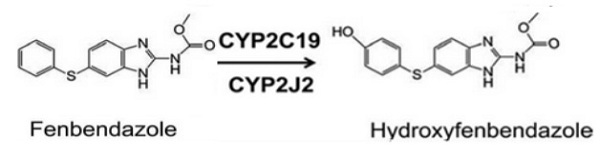
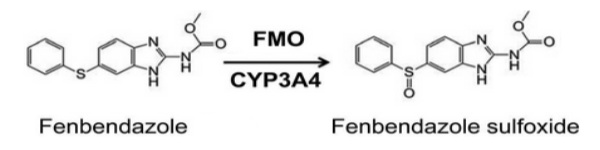
After absorbtion, two different steps of fenbendazole transformation in the body occur – hydroxylation and oxidation.
The first process happens with the help of liver enzymes – CYP2J2 and CYP2C19. Fenbendazole is hydroxylated to form a new metabolite – hydroxyfenbendazole.
The second process is the oxidation of the the sulfide group. This is catalized in the liver by the enzymes CYP3A and flavin-containing monooxygenase. As a result, a new compound forms – oxfendazole (which also displays anticancer and anthelmintic properties).
Interestingly, these findings suggest that fenbendazole is both a drug and prodrug – it leads to a metabolically produced another active drug.
Notably, fenbendazole is not 100% converted to its metabolites due to its poor bioavailability. Only a small amount of fenben is absorbed through our gastrointestinal tract. Ultimately, fenbendazole, together with hydroxyfenbendazole and oxfendazole are excreted with feces.
Keep in mind! One of the enzymes used for metabolizing fenbendazole, CYP2C19, is also responsible for metabolism of drugs like tamoxifen, ebastine, amiodarone, astemizole, mesoridazine, apixaban, thioridazine and cyclosporine.
If a cancer patient decided to take one or several of these drugs together with larger doses of fenbendazole, one should contact her/his medical consultant to get more information about the interactions between fenbendazole and these drugs, possible additional side effects and presumable elevated toxicity.
To find out more visit: https://www.fenbenlab.com/shop/