McGinley Orthopedics, a developer of innovative HandHeld Robotic technology with a goal of changing the standard of medical care, has a new product. The Lever Action Plate System™ is a dynamic, fragment reduction technology that assists surgeons in restoring volar tilt and articular congruity. A novel solution for a wide range of distal radius fractures, The Lever Action Plate System™ provides precise,beam-assisted reduction of intra-articular fractures, creating a structurally sound fixation.
Details for surgeons and industry leaders:
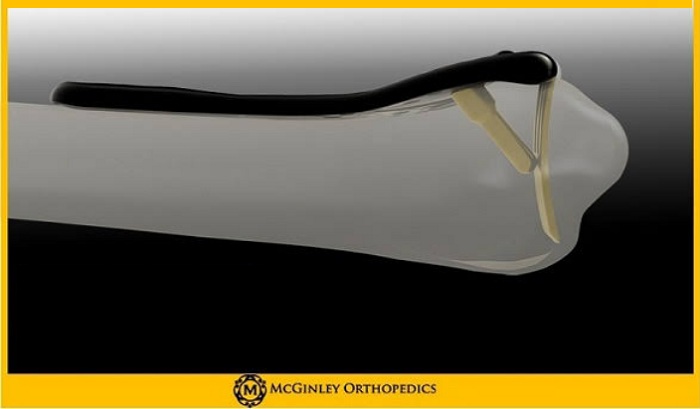
The innovative Lever Action Plate System™features proprietary beams that align volar tilt. The beams are inserted into the nonreduced fracture and with the turn of a screw, the beams and the fracture are elevated into the surgeon’s desired placement. Optimizing alignment, the beams provide independent fixation of the central and radial column. The design’s contoured, subchondral fixation reduces subsidence. An improvement on the current, generic plates available on the market, the system has optional variable angle screws of varying sizes with patented locking technology. The contour of the plate is designed to adhere comfortably to the watershed line on the radius. It is designed to assist the surgeon with restoring volar tilt and in turn, potentially improve patient outcomes. Used in combination with the IntelliSense Drill Technology®, a drill that auto-stops at the far side of the bone and indicates depth measurement for accurate screw sizing, the Lever Action Plate System™ will improve surgical procedures and outcomes.
Why this innovative technology is critical for patients:
The Lever Action Plate System™addresses a real and prevalent need in orthopedics. Distal radius fractures account for about 20%of all fractures.1 in 5 ER treated fractures are of the distal radius. A complication rate of 15% has been reported with traditional volar plating and post-op complication rates can be as high as 80%. The Lever Action Plate System™is an improvement on the standard of care. It is patented and currently pending FDA clearance.