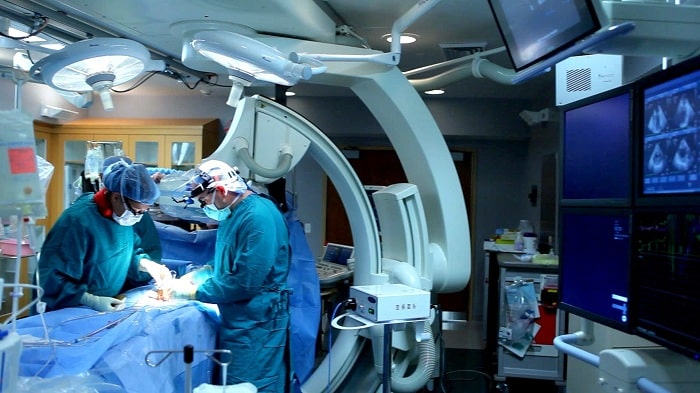
One in ten people in hospital care develops a healthcare-related infection. Together with Getinge, Semcon is developing a new digital platform to make it easier to identify where the risk of infections during surgery is greatest.
In Sweden, infections are the most common form of injury as a result of treatment in hospitals – a problem that is also widespread globally. According to an estimate from the European Centre for Disease Prevention and Control, there are nearly 9 million cases of healthcare-related infections in European hospitals and long-term care every year.
“The digital platform we are currently developing with Getinge helps healthcare better understand a number of evidence-based risk factors. Based on data from the platform, we can develop solutions to reduce the risk of infections in surgical environments. This contributes to reduced human suffering and reduced healthcare costs”, says Johan Kristensson, Team Manager and business responsible connected products at Semcon.
Using a number of sensors in an operating theatre, the digital platform can collect different types of data, which can be displayed in real time as well as analysed afterwards. This creates a better understanding of what is happening in the operating theatre and how to make improvements to reduce healthcare-related infections.
“According to the research, many endogenous and exogenous parameters have an impact, and we want this project to help us evaluate the impact of the environment in the operating theatre. Semcon has extensive experience in both product development and software development, as well as extensive knowledge in medical technology, which makes them a good partner in this project”, says Jonas Andersson, Vice President Global Product Management in Infection Control at Getinge.
A prototype is due to be tested in an operating theatre at a hospital. The project is based on a number of feasibility studies that Semcon has conducted together with Getinge, and Semcon is contributing to the project with excellence in embedded, sensing systems and UX.