Abiomed announces the United States FDA has approved the company’s investigational device exemption application to start an early feasibility study with a first-in-human trial of the 9 French (Fr) Impella ECP™ heart pump. Impella ECP, which stands for expandable cardiac power, will be studied in high-risk percutaneous coronary intervention (PCI) patients.
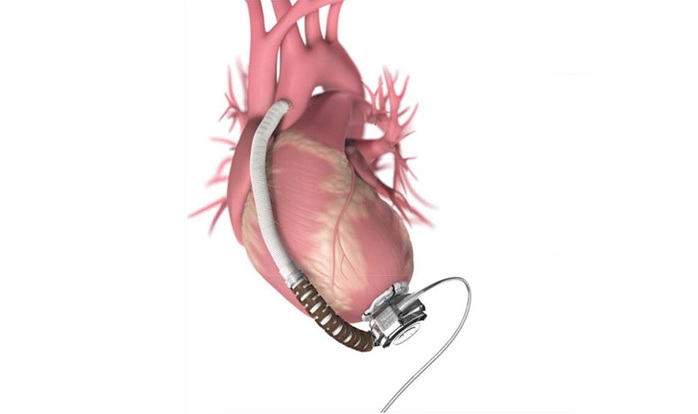
Impella ECP is the world’s smallest heart pump. It achieves peak flows greater than 3.5 L/min and is delivered through a slender-profile sheath. It is un-sheathed in the descending aorta and expands to approximately 18 Fr. Using a specially designed pigtail, it crosses the aortic valve without a wire, and pumps from inside the ventricle. When the procedure is complete, the pump is re-sheathed back down to 9 Fr and removed with that same profile.
A unique design feature of Impella ECP is that it sits across the valve with a soft atraumatic polyurethane cannula that opens only when the pump is flowing. If the pump stops for any reason, this cannula relaxes allowing the valve leaflets to close around it, maintaining valve competency. Impella ECP is intended for short-term mechanical support in patients undergoing a high-risk PCI, to provide both circulatory support and left ventricular unloading.
For interventional cardiologists, vascular access and closure are critical to the success of PCI procedures with mechanical circulatory support. Typically, these procedures require placement of sheaths or cannulas ranging from 13-24 Fr to perform the intervention. Impella ECP allows for a smaller access site, which may increase adoption.
“We are excited about the benefits of this new breakthrough technology, which advances the field of heart recovery,” said Michael R. Minogue, Abiomed’s Chairman, President, and Chief Executive Officer. “Abiomed is making significant investments to develop smaller, smarter, and more connected technology that forms the foundation of the company’s continued leadership in the field of mechanical circulatory support.”
The prospective, multi-center, non-randomized early feasibility study will allow Abiomed, the study investigators, and the FDA to make qualitative assessments about the safety and feasibility of Impella ECP use in high-risk patients. The primary endpoint of the study is successful delivery, initiation and maintenance of adequate hemodynamic support and a composite rate of major device-related adverse events during high-risk PCI. The study protocol will enroll and treat up to five U.S. patients who require revascularization. If successful, enrollment will be expanded to additional patients, pending FDA approval. Study investigators will begin patient enrollment later this calendar year for the first-in-human trial.
Impella ECP is available for investigational use only and is not approved for use outside of clinical studies.
ABOUT IMPELLA HEART PUMPS
The Impella 2.5® and Impella CP® devices are U.S. FDA PMA approved to treat certain advanced heart failure patients undergoing elective and urgent percutaneous coronary interventions (PCI) such as stenting or balloon angioplasty, to re-open blocked coronary arteries. The Impella 2.5, Impella CP, Impella CP with SmartAssist®, Impella 5.0®, Impella LD®, and Impella 5.5™ with Smart Assist® are U.S. FDA approved heart pumps used to treat heart attack or cardiomyopathy patients in cardiogenic shock, and have the unique ability to enable native heart recovery, allowing patients to return home with their own heart. The Impella RP® is U.S. FDA approved to treat right heart failure or decompensation following left ventricular assist device implantation, myocardial infarction, heart transplant, open-heart surgery. The Impella RP is also authorized for emergency use by healthcare providers (HCP) in the hospital setting for providing temporary right ventricular support for up to 14 days in critical care patients with a body surface area ≥1.5 m2, for the treatment of acute right heart failure or decompensation caused by complications related to Coronavirus Disease 2019 (COVID?19), including pulmonary embolism (PE). The Impella RP has neither been cleared or approved for the treatment of acute right heart failure or decompensation caused by complications related to COVID-19. The Impella RP has been authorized for the above emergency use by FDA under an EUA and has been authorized only for the duration of the declaration that circumstances exist justifying the authorization of the emergency use of medical devices under section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the authorization is terminated or revoked sooner.
In Europe, the Impella 2.5, Impella CP and Impella CP with SmartAssist are CE marked for treatment of high-risk PCI and AMI cardiogenic shock patients for up to 5 days. Impella 5.0 and Impella LD are CE marked to treat heart attack or cardiomyopathy patients in cardiogenic shock for up to 10 days. The Impella 5.5™ with Smart Assist® is CE marked to treat heart attack or cardiomyopathy patients in cardiogenic shock for up to 30 days. The Impella RP is CE marked to treat right heart failure or decompensation following left ventricular assist device implantation, myocardial infarction, heart transplant, open-heart surgery, or refractory ventricular arrhythmia.
To learn more about the Impella platform of heart pumps, including their approved indications and important safety and risk information associated with the use of the devices, please visit www.impella.com.
ABOUT ABIOMED
Based in Danvers, Massachusetts, USA, Abiomed, Inc. is a leading provider of medical devices that provide circulatory support. Our products are designed to enable the heart to rest by improving blood flow and/or performing the pumping of the heart. For additional information, please visit: www.abiomed.com.
Abiomed, Impella, Impella 2.5, Impella 5.0, Impella 5.5, Impella LD, Impella CP, Impella RP, SmartAssist and Impella Connect are registered trademarks of Abiomed, Inc., and are registered in the U.S. and certain foreign countries. Impella BTR, Impella ECP, CVAD Study and STEMI DTU Study are pending trademarks of Abiomed, Inc.