Gene therapies have emerged as a promising treatment approach to address various diseases such as cancer, and certain inherited disorders with a single curative dosage. Although they are poised to ensure cost savings in the long run, their high initial expenses and uncertainties regarding their prolonged efficacy are an existing challenge for market players.
Government agencies including, the Food and Drug Administration (FDA), and the European Medicines Agency (EMA), have approved the usage of drug – Kymriah, and Yescarta – to treat elapsed/refractory B-cell Acute Lymphoblastic Leukemia (ALL), and diffuse large B-cell lymphoma (DLBCL). However, the market uptake of both these gene therapy products have been below par.
Despite the ongoing issues, the worldwide revenue of gene therapy market will surpass US$ 5 Bn by 2026, exhibiting a stellar growth rate. At-scale investments in the field of gene-related R&D, and increasing number of late-stage gene therapy candidates in oncology and other genetic disorders are likely to oil the growth engine.
Key Takeaways of the Gene Therapy Market:
- In terms of revenue, Yescarta holds prominence in the gene therapy market, while Luxturna stays ahead in terms of annual growth rate.
- Over half of gene therapy research studies are focused on the field of oncology, attributable to notable rise in caseloads of various cancer types.
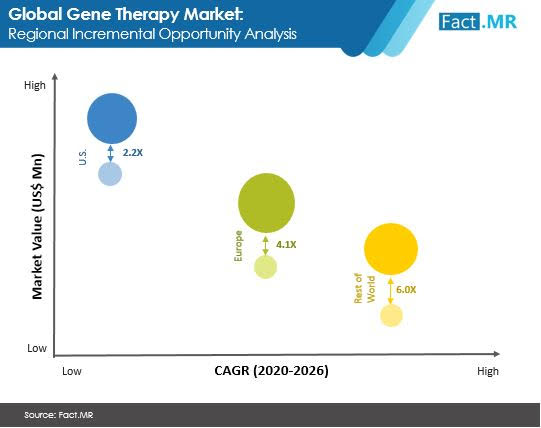
- The US currently represents the most lucrative opportunities in the gene therapy market; while Europe is expected outpace the region towards the end of forecast period.
- Fast uptake of regulatory bodies in the US and Europe for approval and commercialization of gene therapy products are likely to propel market growth.
“Cancer types including, DLBCL, and acute lymphoblastic leukemia (ALL) are among the major causes of mortalities across the globe, especially in developed regions. Realizing the immense potential of gene therapy in treating rare diseases, governments are investing in gene therapy treatment centers to increase access to enhanced patient pool”, Says an Analyst at Fact.MR.
Market Players to Establish Proprietary Manufacturing Units
Amidst rising pool of patients suffering from cancer, governments, biopharmaceutical companies, and research institutes are significantly driving their attention towards gene-centric R&D projects. This is setting high hopes for gene therapy market players. However, as a response to increasing costs and delays associated with pulling in third-party contract manufacturers, leading market players are establishing proprietary manufacturing plants.
For instance, Novartis AG has invested US$ 500 Mn to set up its own gene therapy manufacturing facility, following Pfizer Inc. which has allocated US$ 600 Mn for the same. Such a tactic will help manufacturers safeguard proprietary production techniques and deal with concerns issued by regulatory authorities more efficiently.
As third-party contract manufacturers house limited production capacity for upcoming gene therapy methods under development stage, internal manufacturing units in the long run is an imperative for gene therapy market players.
Key players with strong market presence in global gene therapy market are, but not limited to, Novartis AG, Gilead Sciences Inc., Spark Therapeutics Inc., Sibiono GeneTech Co. Ltd., Spark Therapeutics, Inc., CELGENE CORPORATION, and Orchard Therapeutics Limited, among others.
Looking for more information?
The research study on the gene therapy treatment market by Fact.MR includes an in-depth analysis of major demand-driving factors and trends and a comprehensive evaluation of crucial aspects that are projected to carve the growth of the gene therapy market during 2020–2026. Market statistics have been elucidated based on product (Yescarta, Kymriah, Luxturna, Strimvelis, and Gendicine), application (Ophthalmology, Oncology, and Adenosine Deaminase-deficient Severe Combined Immunodeficiency (ADA-SCID)) and across key regions (The US, Europe and Rest of World).
To Get More Insights on Gene Therapy Treatment Market, Visit- https://www.factmr.com/connectus/sample?flag=S&rep_id=4648





















