Spinal surgery cases have substantially increased over the last few decades due to better technology to diagnose and treat spinal conditions. Surgery often involvessingle to multi-level vertebrae fusions with the use of pedicle hardware for the treatment of deformity, tumor, trauma and degenerative conditions.
Clinical outcomesdepend ona number of factors –accurate selection and sizing of implants, correct placement of hardware, and final alignment of the spine. Pedicle screw misplacement may lead to inadequate biomechanical fixation, poor bone fusion,or neurological or neurovascular injury. Traditional spinal surgery was conducted using intra-operative fluoroscopic systems (C-arms) to guide the placement of pedicle screws.
While these systems greatly facilitated the procedure, they suffered from limitations including image distortion and radiation exposure — predominately to the surgical team. Further, because most C-arms are limited to providing 2 dimensional (2D) projections of the anatomy, the surgeon had to mentally transfer complex 3 dimensional (3D) anatomical detail including pedicle trajectories and their proximity to critical anatomical structures such as the spinal cord or great vessels onto these 2D images.
By the early 2000s, advances in intraoperative 3D imaging technologies (CT and cone-beam CT systems) combined with computer-aided navigation systems were revolutionizing the way these surgeries were performed.
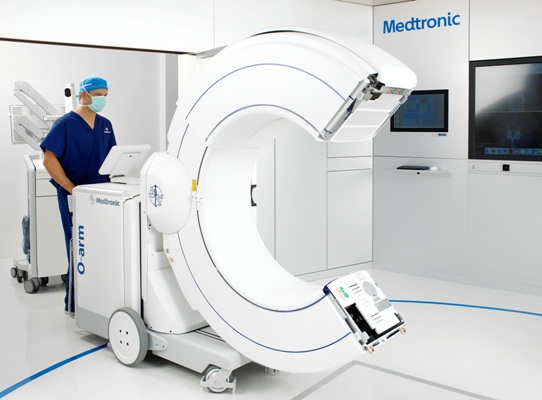
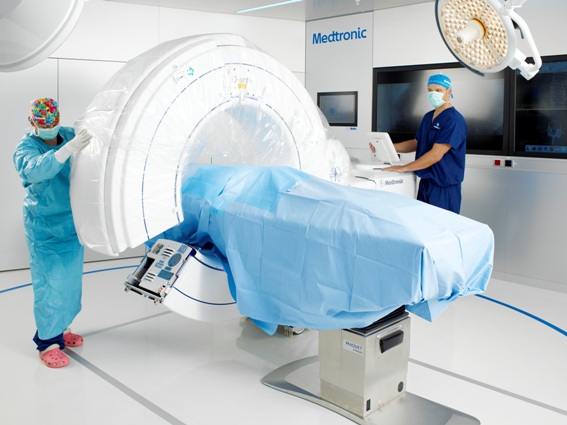
In 2006, Medtronic launched an intraoperative cone-beam CT to the market: theO-arm. Based on flat-panel,solid state x-ray detection technology, the O-arm provides high-resolution fluoroscopic https://www.hhmglobal.com/images as well as 3D volumetric https://www.hhmglobal.com/images. This means more comprehensive imaging and enhanced decision making for surgeons. Driven by these and other key features, such as its breakable gantry and automatic registration for navigation,the O-arm is changing the paradigm for spinal surgery.
This technology has been shown to:
- Increase the accuracy of pedicle screw placement, reducing the likelihood of revisions due to symptomatic, mal-positioned screws. Numerous peer-reviewed studies have demonstrated that surgeons using the O-arm coupled with StealthStation navigation achieve pedicle screw placement accuracy rates between 93% and 100%; compared to rates between 72% and 92% for conventional methods [1-3]
- Reduce surgical complications [1, 5-8]
- Reduce radiation exposure to surgeons and staff [9]
- Provide critical information enabling surgeons to perform more complex surgeries [10-16]
- Empower surgeons to adopt lessinvasive techniques which reduce tissue trauma, the use of medications, and post-operative pain, allof which contribute to speeding patient recovery time
From a hospital economics perspective, these tools supportefficient workflows by supporting the imaging needs of multiple ORs simultaneously. Hospitals can reduce incurred cost associated with revision surgery due to misplaced symptomatic hardware [17]. Costa et al. have estimated a 3.8%cost reduction associated with using the intraoperative imaging (O-arm) and navigation when compared with preoperative imaging and navigation [18].
Others have estimated that at an average re-operation cost of $27,768, the revision cost avoidance translates into monetary savings between $72k and $216k for a case volume of 50 to 150 cases, respectively [19].Furthermore, assurgeons improve their ability to perform minimally invasive procedures, hospitals realize the financial benefits associated with decreased risks of infection, decreased blood loss and reduced hospital stays — translating into savings estimated between $146k and $440k for case volumes of 50 and 150 cases respectively [20-23].
In 2015, Medtronic announced the launch of its second generation O-arm imaging system. Built on the success of the original system, this system added two new main clinical features: a larger3D field of view (40 cm compared with 20 cm) to support stereotactic procedures and pelvic trauma procedures, and a number of lower-dose imaging protocols. The stereotactic feature changes the hospital workflow — hospitals no longer need to coordinate between radiology and the OR to attach and then image the stereotactic localizer. Instead, theO-arm allows all imaging to be performed in the OR, reducing patient and hospital burdens.
The lower-dose imaging protocols provide surgeons with additional options to manage overall patient dosage for the total procedure. This is especially critical in pediatric deformity or in cases that require multiple https://www.hhmglobal.com/images, such as long spinal constructs or stimulation lead placement in the brain.
More than ever, hospitals are under pressure to improve the standard of care, while reducing overall costs. The adoption of these technologies will continue to grow as technology development drives additional value such as improved surgical planning tools and expansion to broader surgical specialties.Finally, future developmentsto integrate surgical robots and navigation technology may enable even more precise localization and real-time execution of surgical plans.
- M. H. Shin et al.: Accuracy and Safety in pedicle screw placement in the thoracic and lumbar spines: Comparison study between conventional C-arm fluoroscopy and navigation coupled with O-arm – guided methods.J Korean Neurosurg Soc.52(3) 204-9. (2012)
- D. Ross and N. Roundy. Accuracy and safety of the placement of thoracic and lumbar pedicle screws using the O-arm intraoperative computed tomography system and Stealth stereotactic guidance. Fed Pract. 30(11) 26-30.(2013)
- Y. Sakai et al.: Segmental pedical screwing for idiopathic scoliosis using computed-assisted surgery. J Spinal Disord Tech. 21(3) 181-186. (2008)
- M. Ammirati and A. Salma. Placement of thoracolumbar pedicle screws using O-arm®-based navigation: Technical note on controlling the operational accuracy of the navigation system. Neurosurg Rev. 36 157-162. (2013)
- N. Luther et al.: Comparison of navigated versus non-navigated pedicle screw placement in 260 patients and 1434 screws: Screw accuracy, screw size, and the complexity of surgery. J Spinal Discord Tech. 28 (5) E298-303. (2015)
- E.W. Nottmeier.A review of image-guided spinal surgery. J Neurosurg Sci.56(1) 35-47. (2012)
- V. Amato et al.: Accuracy of pedicle screw placement in the lumbosacral spine using conventional technique: Computed tomography postoperative assessment in 102 consecutive patients. J Neurosurg: Spine. 12(3) 306-313. (2010)
- S. Yson et al.: Comparison of cranial facet joint violation rates between open and percutaneous pedicle screw placement using intraoperative 3-D CT (O-arm®) computer navigation. Spine.38(4) E251-E258. (2013)
- E. Nottmeier et al.: Surgeon radiation exposure in cone beam computed tomography-based, image-guided spinal surgery.Int J Med Robot.8(2) 196-200.(2012)
- K.M. Scheufler et al.: Percutaneous transforaminal lumbar interbody fusion for the treatment of degenerative lumbar instability. Neurosurgery. 60(4 Suppl2) 203-212. (2007)
- C. Schizas et al.: Minimally invasive versus open transforaminal lumbar interbody fusion: Evaluating initial experience. IntOrthop. 33(6) 1683-1688. (2009)
- Y. Park et al.: Comparison of one-level posterior lumbar interbody fusion performed with a minimally invasive approach or a traditional open approach. Spine. 32(5) 537-543. (2007)
- O. Adogwa et al.: Comparative effectiveness of minimally invasive versus open transforaminal lumbar interbody fusion: 2-year assessment of narcotic use, return to work, disability, and quality of life. J Spinal Disord Tech. 24(8) 479-84. (2011)
- C.W. Penget al.: Clinical and radiological outcomes of minimally invasive versus open transforaminal lumbar interbody fusion. Spine. 34(13) 1385-1389. (2009)
- F. Shunwu F, et al.: Minimally invasive transforaminal lumbar interbody fusion for the treatment of degenerative lumbar diseases. Spine. 35(17) 1615-1620. (2010)
- T.J. Gianaris et al.: Percutaneuous pedicle screw placement with computer-navigated mapping in place of Kirschner wires: Clinical article. J Neurosurg: Spine. 19(5) 608-613. (2013)
- R.G. Watkins et al.: Cost-effectiveness of image-guided spine surgery. Open Orthop J. 4 228-233.(2010)
- F. Costa et al.: Economic study: A cost-effectiveness analysis of an intraoperative compared with a preoperative image-guided system in lumbar pedicle screw fixation in patients with degenerative spondylolisthesis.Spine J.14(8) 1790-6.(2014)
- N. Dea et al.: Economic evaluation comparing intraoperative cone beam CT-based navigation and conventional fluoroscopy for the placement of spinal pedicle screws: a patient-level data cost-effectiveness analysis. Spine J.16(1) 23-31.(2016).
- S. Parker et al.: Minimally invasive versus open transforaminal lumbar interbody fusion for degenerative spondylolisthesis: comparative effectiveness and cost-utility analysis.World Neur. 82 230–8.(2013)
- S. Parker et al.: Post-operative infection after minimally invasive versus open transforaminal lumbar interbody fusion (TLIF): literature review and cost analysis.Minimally Invasive Neurosurgery.54 33–7. (2011)
- K. Phan et al.:Cost–utility of minimally invasive versus open transforaminal lumbar interbody fusion: systematic review and economic evaluation.European Spine Journal. 24.2503-13. (2015)
- R. Toner et al.:Costs to hospitals of acquiring and processing blood in the US: a survey of hospital-based blood banks and transfusion services.Applied health economics and health policy. 9(1) 29–37. (2011)