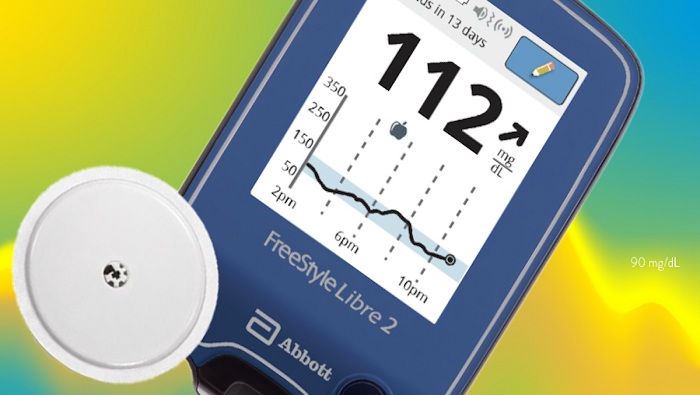
Abbott announced its next-generation, sensor-based glucose monitoring technology, FreeStyle Libre 2, received approval fromHealth Canada for adults and children (4 and older) with diabetes. With new features such as optional, real-time alarms that measure glucose levels every minute, FreeStyle Libre 2 gives users the option to be alerted in real-time of critical events such as hypoglycemia (low glucose levels) or hyperglycemia (high glucose levels).
The wearable technology, which eliminates the need for painful fingersticks, also provides people with diabetes with excellent accuracy and actionable information to better manage their condition, and will be priced at the same cost as the current FreeStyle Libre system.
“For the millions of Canadians with diabetes, Abbott’s next-generation FreeStyle Libre 2 system expands on the life-changing capabilities of our original FreeStyle Libre system with enhanced accuracy, optional alarms and now available for children,” said Marie-Flore Nabor, general manager of Abbott’s diabetes care business in Canada. “This latest technology will transform the way diabetes is currently managed. The FreeStyle Libre 2 is designed to simplify this often complicated-to-manage condition and is accessible and affordable to people with diabetes in Canada.”
As the world leader in sensor-based glucose monitoring, Abbott continues to transform how people with diabetes test their glucose levels. Using Bluetooth technology, the FreeStyle Libre 2 system automatically alerts users when their glucose is high or low without needing to scan the sensor.
The FreeStyle Libre 2 sensor is worn on the back of the upper arm for up to 14 days and measures glucose every minute to help users and their healthcare providers make informed treatment decisions. With a one-second scan using FreeStyle LibreLink, a smartphone app,8 or handheld reader, users can see their glucose reading, trend arrow and eight-hour history. Users can also share data with their physicians or family members via the LibreLinkUp mobile app.
Abbott’s FreeStyle Libre 2 system utilizes the same proprietary wired enzyme technology as the FreeStyle Libre system, which was the first to remove painful fingersticks and is associated with better glucose control, decreased time in hypoglycemia and hyperglycemia, more time in optimal glucose range, and improved HbA1C11.
“Adding an alarm to this glucose sensing technology is definitely a major step to help people with diabetes live more confidently with less fear of high or low glucose levels,” said Dr. Bruce Perkins, M.D., director, Leadership Sinai Centre for Diabetes and Clinician-Scientist, University of Toronto. “We have seen from research with similar technologies that this kind of innovation can improve glucose level control, and even prevent emergency room visits and hospitalizations. For kids and adults alike, it means less pain from fingersticks, much greater insight into patterns, and much more reassurance.”