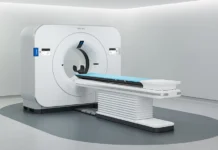
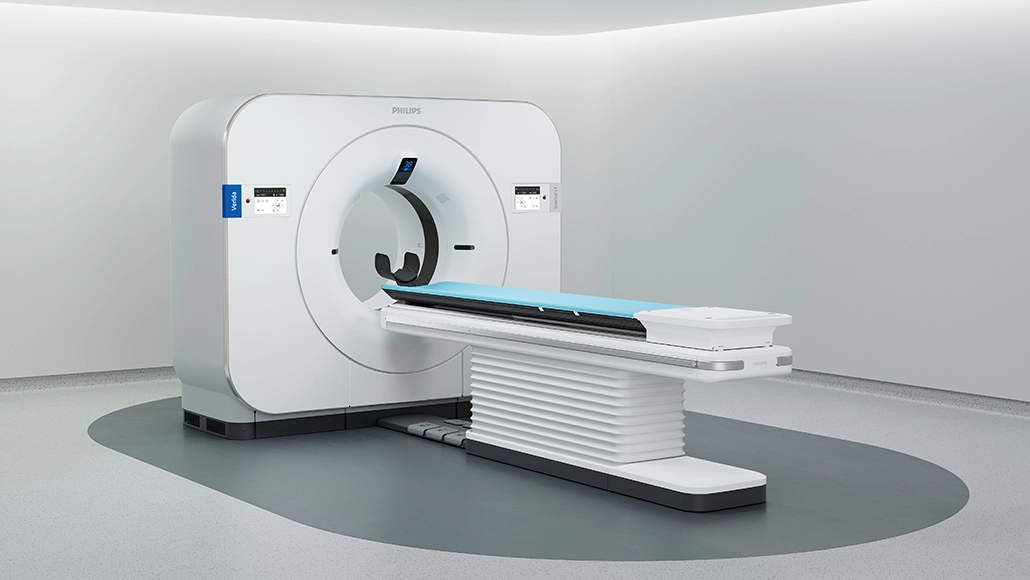
Philips has secured FDA 510(k) clearance for its AI-powered Spectral CT Verida system, marking a significant step in advancing imaging capabilities. Introduced in December 2025, the platform is positioned as the first detector-based spectral CT system fully powered by artificial intelligence. The development reflects the company’s push to integrate AI across diagnostic imaging processes, aiming to enhance performance and clinical usability.
The system has been engineered to optimize the entire imaging workflow, from acquisition to reconstruction. By leveraging AI, it reduces system noise, improves image clarity, and accelerates clinical processes. Designed for seamless integration, the platform delivers PACS-native outputs and embeds directly into existing workflows. Spectral imaging enables the analysis of how tissues absorb varying X-ray energy levels, allowing detector-based systems to generate multiple spectral outputs from a single scan without compromising scan time or performance.
The technology is intended for broad clinical application, including radiology, interventional radiology, and cardiology. It also supports oncology workflows, particularly in treatment preparation and radiation therapy planning. The system combines high-definition conventional imaging with advanced spectral capabilities, while AI-driven reconstruction delivers enhanced image quality with reduced noise. Additionally, the platform is designed to lower radiation dose without affecting diagnostic output and can cut energy consumption by up to 45%.
Powered by third-generation Nano-panel Precise dual-layer detector technology, the system significantly improves processing efficiency. It can reconstruct up to 145 images per second and complete full exams in under 30 seconds, enabling throughput of up to 270 exams daily. This represents a twofold speed increase compared to earlier models. The platform builds on proprietary Spectral Precise Imaging technology, incorporating deep learning to refine imaging outcomes.
Dan Xu, Business Leader of CT at Philips, said:
“With FDA clearance for Verida, we are bringing the next evolution of spectral CT to more markets. By combining always-on spectral imaging with AI-powered reconstruction, Verida enables clinicians to see more, first time right, supporting faster, more informed decisions and expanding the role of CT across clinical pathways.”