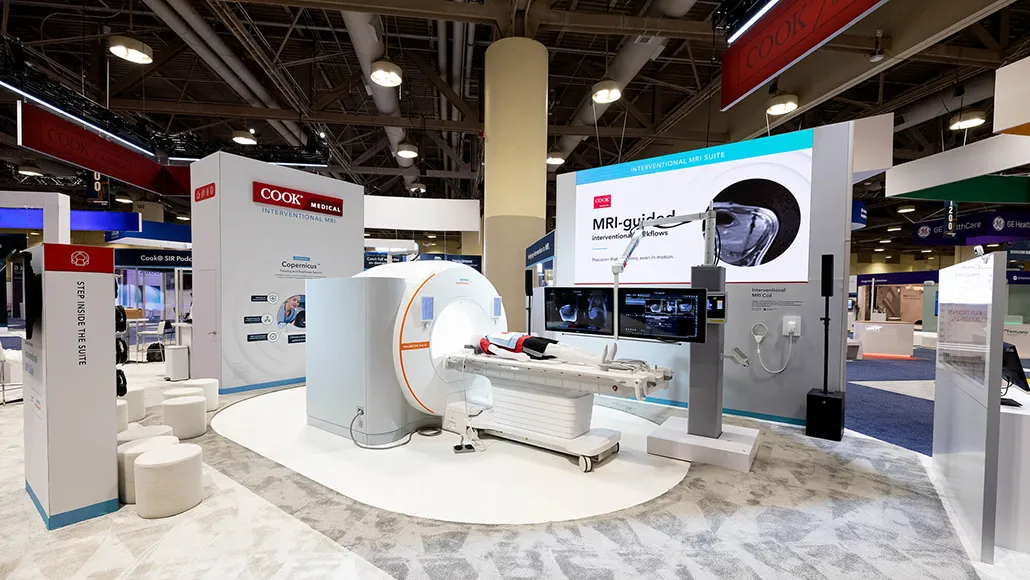
Siemens Healthineers and Cook Medical have introduced a fully integrated interventional magnetic resonance imaging suite, marking a new phase in their collaboration on MRI-guided procedures. The announcement was made at the Society of Interventional Radiology (SIR) 2026 Annual Scientific Meeting, where both companies showcased the system designed to support minimally invasive interventions without the use of ionizing radiation. This MRI Suite launch represents a significant step forward in combining imaging, devices, and workflow systems into a single clinical environment.
The integrated solution brings together real-time MRI capabilities with MR-conditional interventional tools and structured clinical workflows. It builds on a partnership formalised in September 2025, when the two companies aligned their expertise—pairing Siemens Healthineers’ imaging technologies with Cook Medical’s procedural specialisation. The system incorporates devices specifically developed for MRI environments alongside the Magnetom Free.XL platform, creating a unified framework for interventional imaging and treatment delivery.
Within the suite, multiple components are integrated to streamline clinical use and expand procedural capabilities:
- Advanced imaging technology designed for real-time guidance during interventions
- Purpose-built interventional devices tailored for MRI compatibility
- Suite planning support and clinical education resources
- Integration with SIR VIRTEX, a clinical data registry and analytics platform
- Continuous data feedback to enhance procedural learning and outcomes
The companies state that this MRI Suite launch enables physicians to perform procedures that benefit from enhanced soft-tissue visibility while avoiding radiation exposure. Applications include oncology and soft-tissue interventions, with use cases spanning biopsies, ablations, and procedures in pediatric and cardiac care. According to Peter Polverini III, VP of Cook Medical’s iMRI division, “The iMRI Suite is more than a technology; it’s a fundamental shift in how we approach intervention. By bringing together advanced imaging, purpose-built devices, and integrated workflows, we are enabling physicians to see more, treat with greater precision, and ultimately deliver better outcomes without exposing patients or care teams to radiation.”
Pete Yonkman, president of Cook Medical and Cook Group, added: “MRI offers unique advantages for image-guided intervention, particularly for procedures involving soft tissue. Through our collaboration with Siemens Healthineers and leading clinicians, we are working to advance the development of radiation-free interventional approaches.” Andreas Schneck, head of Magnetic Resonance at Siemens Healthineers, said: “At Siemens Healthineers, it is our goal to elevate health globally. Magnetom Free.XL is designed to unlock the full potential of MR in the interventional suite, expanding the imaging toolbox by matching the right modality to the right patient at the right time.”