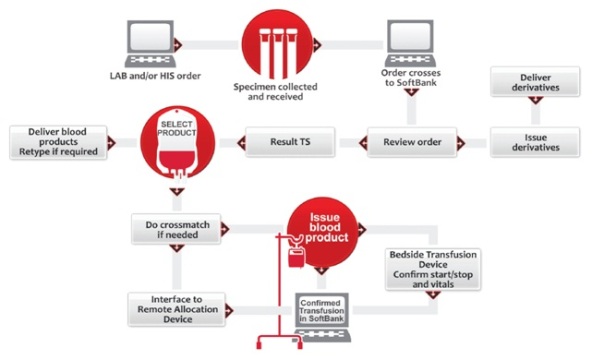
An overview of imporant patient safety benefits and workflow advantages of the integrated single database solution for blood bank transfusion management and administration from SCC Soft Computer.
According to a 2007 report by the World Health Organization, patient misidentification was cited in more than 100 individual root cause analyses by the United States Department of Veterans Affairs National Center for Patient Safety from January 2000 to March 2003.
The major areas where patient misidentification can occur include drug administration, phlebotomy, blood transfusions, and surgical procedures. A mislabeled blood specimen could be catastrophic to the patient and could result in a patient receiving a transfusion of the wrong blood type, a misdiagnosis, or incorrect medical treatment.
In today’s fast-paced, highly regulated transfusion service setting, it is important to provide safe products to accommodate the diverse needs of a complex multisite facility, as well as the highly specialized needs of a single site facility.
The SoftBank® / SoftID.Tx Advantage A better workflow for transfusion administration
SoftBank®, SCC Soft Computer’s Blood Bank Transfusion Service Information Management System, provides critical safety functions throughout the system, allowing users to focus on what’s important: providing safe blood components, tissue, and derivative products to their patients.
SoftBank provides a wide range of exceptional features that streamline many of the routine, manual, and time-consuming tasks associated with blood bank and blood donor protocols. These timesaving improvements translate into revenue for transfusion services departments of all sizes. SCC is one of the few LIS vendors who can say that their systems work as well in a 150-bed community hospital as they do in a high-volume healthcare system. SCC’s robust integrated information systems dramatically increase productivity in the laboratory enabling hospitals of all sizes to optimize their workflows.
With high-performance functionality and robust features, this enhanced laboratory blood bank information system also provides a comprehensive and cost-effective centralized pre-transfusion testing and inventory management software solution. The database management system offers fast, direct linking between records and transactions, dramatically increasing levels of security and integrity when performing and documenting activities and events.
SoftBank has received FDA 510(k) clearance as a Class II Medical Device and provides a controlled and documented process for blood bank testing and issue of products, thereby reducing risk of improper actions or products being administered.
With the versatility of SCC’s SoftID.Tx [also FDA 510(k) cleared], users can add positive patient identification (PPID) into their existing workflows. By leveraging the power of the integrated SoftBank/SoftID.Tx platform, blood banks and transfusion services can streamline their processes. The parameter-driven setup allows the builder to create a workflow perfectly suited to the users’ routines, or to create a totally new and better-suited workflow to make full use of all the features included, or something in between. The design is up to the individual client.
Unlike stand-alone transfusion administration systems or HIS/EMR vendor-provided transfusion administration systems, SCC’s SoftID.Tx is a fully integrated module of SCC’s SoftBank blood bank and transfusion service information management system. Because SoftID.Tx is part of SoftBank, it yields important patient safety and workflow advantages that make it the overwhelming choice of SCC’s SoftBank clients who have elected to implement a PPID solution for transfusion administration.
Support bedside transfusion administration and bedside collection of patient vitals during the blood transfusion process, eliminate manual entry, and automate workflow with the fully integrated SoftBank/SoftID.Tx solution.
The U.S. FDA has granted 501(k) clearance to SCC’s SoftBank version 25.5 (BK140128 08/05/2014).
The U.S. FDA has granted 501(k) clearance to SCC’s SoftID.Tx version 1.0 (BK090078 04/28/2010).
According to Gilbert Hakim, SCC Soft Computer CEO, “SoftBank is a centralized transfusion service management system, designed and developed in 1992. The first version, released in 1993, provided a comprehensive, cost-effective pre-transfusion testing and inventory management solution for SCC SoftLab clients and those using other HIS/LIS solutions. Over the years, SoftBank has evolved to keep pace with regulatory changes and with the addition of interfaced instrumentation. Two reasons for the product’s success are SCC’s commitment and funding for steady and constant improvement of SoftBank for more than 20 years, and input from our dynamic client base.”