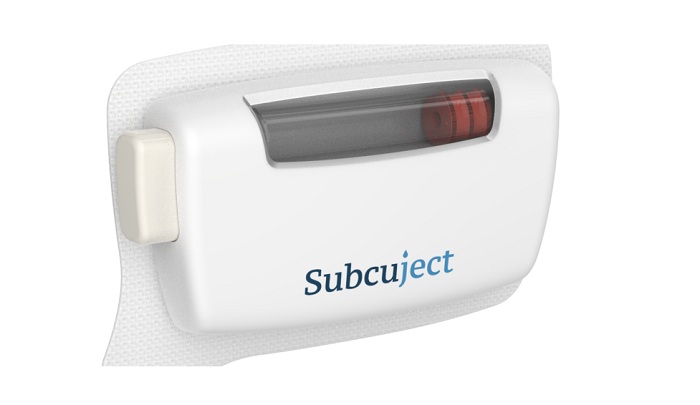
Phillips-Medisize, a Molex company, and leader in the design, development, and manufacturing of drug delivery, diagnostic, and MedTech devices, collaborates with Subcuject, a global innovator of proprietary device platforms, to bring a ground-breaking wearable bolus injector to market. The On-Body Delivery System technology, based on osmosis, is now available for the next stage of drug-specific wearable injector solution development following Subcuject’s intellectual property filing of the concept.
“We are very pleased to collaborate with Phillips-Medisize to complete the development and manufacturing needed to bring our product innovation to pharma customers successfully,” said Jesper Roested, CEO of Subcuject. “Together, we can address the emerging need for inexpensive, prefilled, and single-use injectors that can deliver drugs at about 1mL per minute.”
The new Wearable Osmotic Bolus Injector is designed to be a low-cost, patient-friendly, prefilled wearable injector for single-use. The product uses osmosis to generate the force to complete a full injection cycle, requiring minimal components, which reduces development time and costs. Moreover, no additional electronics or batteries are required, resulting in further cost reductions and a lower environmental impact. Subcuject’s management team and board have decades of experience and a track record in MedTech, pharma, and drug delivery.
“Teaming with Subcuject to develop an affordable, versatile wearable injector leverages our combined strengths and global expertise in proprietary device platforms,” said Paul Chaffin, president of Phillips-Medisize. “We’re excited to develop the technology and pave the way toward commercialization.”
Additionally, Subcuject and Phillips-Medisize are exploring the opportunity to meet the growing demand for larger-volume drug delivery. The device will enable patients to self-administer certain medications almost anywhere, instead of requiring healthcare professionals to do so in a clinical setting.
“The urgent demands for quality and affordable patient care are driving momentum across the entire drug-delivery sector, including the wearable injector segment,” said Paul Jansen, a leading drug-device consultant and Subcuject board member. “The collaboration between Phillips-Medisize and Subcuject combines technology innovation, proven engineering, and manufacturing expertise to speed and scale the realization of this exciting new product.” Founded in 2017, Denmark-based Subcuject is a technology development company focused on developing an innovative and proprietary device platform for wearable bolus injection. The company is backed by Danish venture funds VF Venture and Capnova.
Phillips-Medisize: End-to-End Expertise
For more than 80 years, Phillips-Medisize has engaged with leading Healthcare and Life Science companies to develop innovative products that help people live healthier, more productive lives. The company’s unique end-to-end capabilities, from design and development, to global manufacturing, allow Phillips-Medisize to accelerate time to market while driving significant cost efficiencies. On average, Phillips-Medisize commercializes 50 new products a year for customers, including the first-to-market FDA-registered drug-delivery device utilizing a connected health system.