According to new report titled, “Ophthalmic Surgical Devices and Therapeutics Market – Global Industry Analysis, Size, Share, Growth, Trends, and Forecast, 2018•2026” the ophthalmic surgical devices and therapeutics market is expected to reach at US$ 43,794.4 Mn by the end of 2018. The market is anticipated to reach US$ 68,898.4 Mn by 2026 and expand at a CAGR of 5.8% from 2018 to 2026.
Rise in number of geriatric patients, increase in awareness regarding eye disorders, and high unmet medical needs are expected to augment the global market from 2018 to 2026. The ophthalmic surgical devices and therapeutics market is projected to expand owing to an increase in the prevalence of eye-related disorders among the population, demand for better treatments, and developing healthcare infrastructure in various countries across the globe.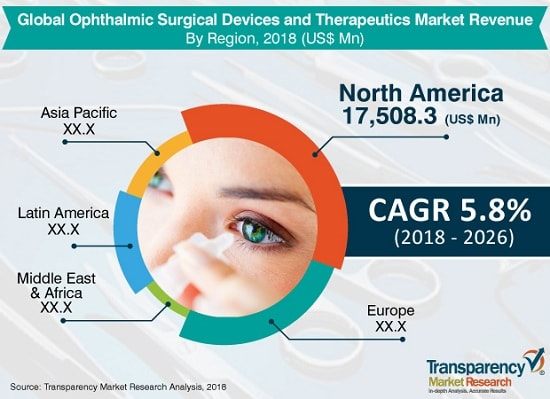
Increasing geriatric population and rising awareness regarding eye diseases
According to data published by the United Nations Department of Economics and Social Affairs, the geriatric population is expected to double by the end of 2050 and is projected to reach nearly 2.1 billion. This increasing geriatric population is prone to significant risk of eye-related disorders, including blindness. Public awareness regarding common eye conditions is increasing in developing countries, leading to early diagnosis and treatment of eye diseases. Furthermore, there are a large number of organizations dedicated to fight blindness, restore vision, and create awareness regarding eye health. National Eye Institute, National Association for Visually Handicapped, National Federation of the Blind, and Prevent Blindness are some eye health organizations concerned with prevention of eye diseases.
Increase in incidence rates of eye related disorders
According to the World Health Organization (WHO), there are 285 million visually impaired people globally, approximately 90% of them reside in low to middle income countries. Furthermore, approximately 95 million people suffer from cataract, and 20 million suffer from various eye conditions, including glaucoma, muscular degeneration, infections, and childhood-related conditions. Significant rise in incidence of eye-related diseases, especially in developed countries, is anticipated to drive the ophthalmic surgical devices and therapeutics market between 2018 and 2026. Patients undergoing open angle glaucoma and age related macular degeneration (AMD) in the U.S., were 2.7 million and 2.1 million, respectively. This number is projected to increase to 3.3 million and 2.5 million, respectively, by 2020. Increasing patient pool in developing countries as well as developed countries is estimated to propel healthcare spending for eye treatments in these countries and drive the ophthalmic surgical devices and therapeutics market.
High unmet needs for refractive corrections
According to an article published by the British Journal of Visual Impairment, refractive errors are the leading cause of visual impairment and at the same time, most treatable cause of visual impairment in children. Furthermore, according to this article, more than 90% of people with refractive errors reside in low-income and poor countries and cost is a major obstacle to accessing glasses and meeting the need for correction. Studies indicate that rates of willingness to pay for glasses are low, and cost and affordability is a primary reason for not using glasses in China, India, countries in Africa, and several developing countries across the globe.
Therapeutics segment dominates the global market due to its choice as first line of treatment for ocular diseases
In terms of product type, therapeutics is a highly attractive segment of the global ophthalmic surgical devices and therapeutics market, followed by surgical devices and vision care segments. This is attributable to the preference for drugs as the first line of treatment for any eye-related diseases. Expansion of the segment is primarily attributed to the increased prevalence of glaucoma and ocular infections among the population. According an article published by the American Academy of Ophthalmology, the global prevalence of glaucoma for population aged 40 to 80 years is 3.54%. The prevalence of primary open-angle glaucoma is considerably high in Africa, i.e., 4.2%, and the prevalence of primary angle-closure glaucoma (PACG) is significantly high in Asia, i.e., 1.09%.
North America dominates the global market owing to significant technological advancements in the region
North America dominates the global ophthalmic surgical devices and therapeutics market due to a large patient pool, high cost of specialty branded drugs, high cost of ocular surgeries, and increase in the geriatric population in the region. The region is estimated to maintain its dominance during the forecast period. According a U.S eye disease statistics, published by the American Academy of Ophthalmology, cataract, age-related macular degeneration, glaucoma, and diabetic retinopathy are the most common causes of visual impairment among the population in the U.S. Additionally, approximately 7.32 million people in the U.S. are expected to suffer from primary open-angle glaucoma by 2050. Moreover, rising healthcare expenditure and increasing investments are key factors that are anticipated to boost the ophthalmic surgical devices and therapeutics market in the next few years. The ophthalmic surgical devices and therapeutics market in Asia Pacific is projected to expand at a notable CAGR due to increasing awareness regarding eye diseases in developing countries and government initiatives introduced in the region.
Investments by key players is driving the global ophthalmic surgical devices and therapeutics market.
Key players dominating the ophthalmic surgical devices and therapeutics market include Allergan Plc., Johnson & Johnson Services, Inc., F. Hoffmann-La Roche Ltd., Novartis AG, Bausch Health Companies Inc., Regeneron Pharmaceuticals, Inc., Santen Pharmaceutical Co., Ltd., Carl Zeiss Meditec AG, HOYA Corporation, and Bayer AG.
Visit More Information: https://www.researchmoz.us/enquiry.php?type=S&repid=2172750




















